Abstract
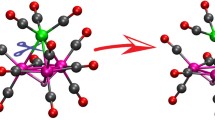
Electrochemical studies show that it is possible to move step-wise and reversibly between the redox congeners of the series [Fe3Pt3(CO)15]n,n=2-/1-/0. By contrast, a multielectron reduction of the dianion leads to an irreversible demolition of the species. When [Fe3Pt3(CO)15]2− is treated with one or two equimolar amounts of the oxidant [Fe(C5H5)2]+, the oxidized species (n=1- andn=0) can be also obtained. It can be established or extrapolated from the already known structures of the dianion and the monoanion that the successive oxidations strengthen the inner Pt-Pt linkages of the overall quasiplanar Fe3Pt3 skeleton. MO analysis, by establishing the antibonding nature of the frontier level from which the electrons are added or subtracted, allows the correlation of the bonding features of the inner Pt3 skeleton with the redox propensities of the system.
Similar content being viewed by others
References
G. Longoni, M. Manassero, and M. Sansoni (1980).J. Am. Chem. Soc. 102, 7974.
C. Mealli (1985).J. Am. Chem. Soc. 107, 2245.
N. J. Taylor, P. Chieh, and A. J. Carty (1975).J. Chem. Soc. Chem. Comm. 1975, 448.
R. Bender, P. Braunstein, A. Tiripicchio, and M. Camellini (1985).Angew. Chem. Int. Ed. Engl. 24, 861.
G. W. Bushnell, K. R. Dixon, P. M. Moroney, A. D. Rattray, and C. Wan (1977).J. Chem. Soc. Chem. Comm. 1977, 709.
D. E. Berry, G. W. Bushnell, K. R. Dixon, P. M. Moroney, and C. Wan (1985).Inorg. Chem. 24, 2625.
S. M. Owen (1988).Polyhedron 7, 253.
M. Green, J. A. K. Howard, M. Murray, J. L. Spencer, and F. G. A. Stone (1977).J. Chem. Soc. Dalton Trans. 1977, 1509.
D. C. Moodey and R. R. Ryan (1977).Inorg. Chem. 16, 1052.
M. J. S. Dewar (1984).J. Am. Chem. Soc. 106, 669.
C. Mealli and D. M. Proserpio (1989).Comments Inorg. Chem. 9, 37.
C. Mealli and D. M. Proserpio (1990).J. Am. Chem. Soc. 112.
D. M. Duggan and D. N. Hendrickson (1975).Inorg. Chem. 14, 955.
E. R. Brown and J. R. Sandifer,in B. W. Rossiter and J. F. Hamilton (eds.),Physical Methods of Chemistry: Electrochemical Methods (Wiley, New York, 1986), Vol. 2, Ch.4.
W. E. Geiger (1985).Prog. Inorg. Chem. 33, 275.
R. Hoffmann and W. N. Lipscomb (1962).J. Chem. Phys. 36, 2179, 3489.
R. Hoffmann (1963).J. Chem. Phys. 39, 1397.
R. Hoffmann, H. Fujimoto, J. R. Swenson, and C.-C. Wan (1973).J. Am. Chem. Soc. 95, 7644.
R. Hoffmann and H. Fujimoto (1974).J. Phys. Chem. 78, 1167.
T. A. Albright, J. K. Burdett, and M. H. Whangbo,Orbital Interactions in Chemistry (Wiley, New York, 1985).
R. C. Adams, G. Chen, and J.-G. Wang (1989).Polyhedron 8, 2521.
R. R. Gagné, C. A. Koval, T. J. Smith, and M. C. Cimolino (1979).J. Am. Chem. Soc. 101, 4571.
L. Casella, M. Gullotti, A. Pintar, F. Pinciroli, R. Viganò, and P. Zanello (1989).J. Chem. Soc. Dalton Trans. 1989, 1161.
C. Mealli and D. M. Proserpio (1990).J. Chem. Ed. 67, 399.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Pergola, R.D., Garlaschelli, L., Mealli, C. et al. Electrochemistry of the two-dimensional heteronuclear [Fe3Pt3(CO)15]n clusters (n=2-, 1-, 0): MO treatment of the skeletal adjustments in 86-84e − congeners. J Clust Sci 1, 93–106 (1990). https://doi.org/10.1007/BF00703587
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00703587