Abstract
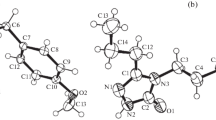
The title compounds were prepared and X-ray analysis was performed (R = 0.064 and 0.035 for 1309 and 1637 reflections, respectively). In the former the intramolecular S→Si coordination interactions are absent and the Si atom has undistorted tetrahedral coordination with an S...Si distance of 4.034 Å. Crystals of the latter are built of centrosymmetric dimeric H-complexes (S...H(N) 2.58 Å, the S...HN angle 168°).
Similar content being viewed by others
References
Yu.I.Baukov, E.P.Kramarova, A.G.Shipov, G.I.Oleneva, O.B.Artamkina, A.I.Albanov, M.G.Voronkov, and V.A.Pestunovich,Zh.Obshch.Khim., 1989,59, 127 [J.Gen.Chem. USSR, 1989.].
I.D.Kalikhman, A.I.Albanov, O.B.Bannikova, L.I.Belousova, M.G.Voronkov, V.A.Pestunovich, and A.G.Shipov, Yu.I.Baukov,J.Organometal.Chem., 1989,361, 147.
M.G.Voronkov, A.I.Albanov, A.E.Pestunovich, V.N.Sergeev, S.V.Pestunovich, I.I.Kandror, and Yu.I.Baukov,Metallorgan. Khim., 1988,1, 1435 [Organomet.Chem.].
J.Tafel and P.Zavaczeck,Ber., 1907,40, 2842.
A.Bondi,J.Phys.Chem., 1964,68, 441.
A.A.Macharashvili, Yu.E.Ovchinnikov, Yu.T.Struchkov, V.N.Sergeev, S.V.Pestunovich, and Yu.I.Baukov,Izv.Akad. Nauk, Ser.Khim. 1993, 189 [Rus.Chem.Bull., 1993,42, 173].
F.H.Allen, O.Kennard, D.G.Watson, L.Brammer, A.G.Orpen, and R.Taylor,J.Chem.Soc.Perkin Trans.2, 1987, 1.
A.A.Macharashvili, V.E.Shklover, Yu.T.Struchkov, G.I.Oleneva, E.P.Kramarova, A.G.Shipov, and Yu.I.Baukov,J.Chem.Soc., Chem.Commun., 1988, 684.
W.Robinson and G.M.SheldrickSHELX. In:Crystallographic Computing-Techniques and New Technologies. Eds. N.W.Isaacs, M.RTaylor, Oxford, Oxford Univ.Press, 1988, 366.
R.G.Gerr, A.I.Yanovsky, and Yu.T.Struchkov,Kristallografiya, 1983,28, 1029 [Crystallography].
Author information
Authors and Affiliations
Additional information
Translated fromIzvestiya Akademii Nauk, Seriya Khimicheskaya, No. 1, pp. 198–202, January, 1993.
Rights and permissions
About this article
Cite this article
Mozzhukhin, A.O., Ovchinnikov, Y.E., Antipin, M.Y. et al. Crystal and molecular structure of 1-(trimethylsilylmethyl)hexahydroazepine-2-thione and hexahydroazepine-2-thione. Russ Chem Bull 42, 181–184 (1993). https://doi.org/10.1007/BF00700005
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00700005