Summary
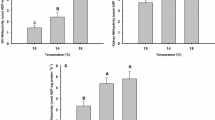
Concentrations of lactic acid in bodies of inactive neotenic tiger salamanders were similar to those recorded for inactive transformed animals. Transformed salamanders swimming against a water current for 5 min accumulated significantly more lactate in their body fluids than did neotenic salamanders, but subsequently metabolized the lactate at higher rates than characterized neotenic animals. Total body lactate in both morphs decreased rapidly in the first 1–1/2 h after activity, and within 6 h had returned to the level (common to both morphs) observed prior to activity. Whereas neotenic animals apparently swim with greater efficiency than transformed salamanders (4.26 μmole ATP/gvs. 6.06 μmole ATP/g expended by neotenic and transformed animals, respectively, during 5 min of continuous swimming), the transformed animals exhibit a metabolic adaptation to aquatic existence enabling them rapidly to recover from a lactacid debt.
Similar content being viewed by others
References
Abbott, B.C., Brady, A.J.: Amphibian muscle. In: Physiology of the Amphibia (ed. J.A. Moore), pp. 329–370. New York: Academic Press 1964
Bennett, A.F., Licht, P.: Anaerobic metabolism during activity in lizards. J. comp. Physiol.81, 277–288 (1972)
Bennett, A.F., Licht, P.: Relative contributions of anaerobic and aerobic energy production during activity in Amphibia. J. comp. Physiol.87, 351–360 (1973)
Bennett, A.F., Licht, P.: Anaerobic metabolism during activity in amphibians. Comp. Biochem. Physiol.48A, 319–327 (1974)
Bennett, A.F., Wake, M.H.: Metabolic correlates of activity in the caecilianGeotrypetes seraphini. Copeia1974, 764–769 (1974)
Black, E.C., Robertson, A.C., Parker, R.R.: Some aspects of carbohydrate metabolism in fish. In: Comparative physiology of carbohydrate metabolism in heterothermic animals (ed. A.W. Martin), pp. 89–124. Seattle: Univ. Washington Press 1961
Brattstrom, B.H.: A preliminary review of the thermal requirements of amphibians. Ecology44, 238–255 (1963)
Carmer, S.G., Swanson, M.R.: Evaluation of ten pairwise multiple comparison procedures by Monte Carlo methods. J. Amer. stat. Assoc.68, 66–74 (1973)
Dent, J.N.: Survey of amphibian metamorphosis. In: Metamorphosis (eds. W. Etkin and L. I. Gilbert), pp. 271–312. New York: Appleton-Century-Crofts 1968
Dixon, W.J., Massey, F.J., Jr.: Introduction to statistical analysis. New York: McGraw-Hill Book Co. 1969
Guimond, R.W., Hutchison, V.H.: The effect of temperature and photoperiod on gas exchange in the leopard frog,Rana pipiens. Comp. Biochem. Physiol.27, 177–195 (1968)
Heath, A.G.: Behavioral thermoregulation in high altitude tiger salamanders,Ambystoma tigrinum. Herpetologica31, 84–93 (1975)
Hutchison, V.H., Turney, L.D.: Glucose and lactate concentrations during activity in the leopard frog,Rana pipiens. J. comp. Physiol.99, 287–295 (1975)
Kenney, J.W., Rose, F.L.: Oxygen requirements and activity rhythms of the tiger salamander,Ambystoma tigrinum (Amphibia: Caudata). Herpetologica30, 333–337 (1974)
Jackson, D.C., Silverblatt, H.: Respiration and acid-base status of turtles following experimental dives. Amer. J. Physiol.226, 903–909 (1974)
Prange, H.D., Schmidt-Nielsen, K.: The metabolic cost of swimming in ducks. J. exp. Biol.53, 763–777 (1970)
Prosser, C.L.: Muscles. In: Comparative animal physiology (ed. C.L. Prosser), pp. 719–788. Philadelphia: W.B. Saunders Co. 1973
Seymour, R.S.: Physiological correlates of forced activity and burrowing in the spadefoot toad,Scaphiopus hammondii. Copeia1973, 103–115 (1973)
Turney, L.D., Hutchison, V.H.: Metabolic scope, oxygen debt and the diurnal oxygen consumption cycle of the leopard frog,Rana pipiens. Comp. Biochem. Physiol.49A, 583–601 (1974)
Vinegar, A., Hutchison, V.H.: Effect of photoperiod on pulmonary and cutaneous gas exchange in the green frog,Rana clamitans. Zoologica50, 47–53 (1965)
Wade, M., Rose, F.L.: A comparison of the hemoglobins of larval and transformedAmbystoma tigrinum. Copeia1972, 889–892 (1972)
Webb, R.G., Roueche, W.L.: Life history aspects of the tiger salamander (Ambystoma tigrinum mavortium) in the Chihuahuan Desert. Great Basin Nat.31, 193–212 (1971)
Whitford, W.G., Hutchison, V.H.: Effect of photoperiod on pulmonary and cutaneous respiration in the spotted salamander,Ambystoma maculatum. Copeia1965, 53–58 (1965)
Whitford, W.G., Sherman, R.E.: Aerial and aquatic respiration in axolotl and transformedAmbystoma tigrinum. Herpetologica24, 233–237 (1968)
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Cushman, J.R., Packard, G.C. & Boardman, T.J. Concentrations of lactic acid in neotenic and transformed tiger salamanders (Ambystoma tigrinum) before and after activity. J Comp Physiol B 112, 273–281 (1976). https://doi.org/10.1007/BF00692298
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00692298