Summary
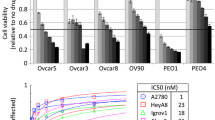
Adozelesin is a derivative of an extremely cytotoxic compound, CC1065. This entirely new class of drug binds preferentially to DNA and facilitates alkylation reaction. In the present study, we used the adenosine triphosphate (ATP) chemosensitivity assay to compare the cytotoxic potency of Adozelesin with that of common chemotherapeutic agents in ten gynecologic-cancer cell lines. Flow cytometry was also used to study its effects on cell-cycle kinetics. The mean drug concentrations required to produce a 50% reduction in ATP levels as compared with controls [IC50] were: Adriamycin, 0.17±0.06 μm; 4OH-Cytoxan, 18±3 μm; cisplatin, 17±7 μm; 5-fluorouracil, 183±116 μm; and Adozelesin, 11.0±5.4pm. Thus, Adozelesin was 104–107 times more potent than Adriamycin, cisplatin, 5-fluorouracil, and Cytoxan. Cell kinetics studies revealed significant S and G2 blocks such as those previously reported for other alkylating agents.
Similar content being viewed by others
References
Alberts D, Chen G (1980) Tabular summary of pharmacokinetic parameters revelant to in vitro drug assay. In: Salmon S (ed) Cloning of human tumor stem cells. Alan J. Liss, New York, p 351
Alberts D, Elnspahr J, Sipes IG (1989) In vitro cytotoxicity of a novel DNA minor groove binding anticancer agent (U-73975) against doxorubicin sensitive and resistant human tumor cell lines. Proc Am Assoc Cancer Res 30: A1957
Anai H, Maehara Y, Kusumoto H, Sugimachi K (1987) Comparison of the chemosensitivity of human neoplastic tissues between succinate dehydrogenase inhibition test and ATP assay. Clin Chim Acta 166(1): 107
Bertelsen CA, Sondak VK, Mann BD, Korn EL, Kern DH (1984) Chemosensitivity testing of human solid tumors. A review of 1582 assays with 258 clinical correlations. Cancer 53: 1240
Bhuyan BK, Crampton SL, Adams EG (1983) Cell cycle effects of CC-1065. Cancer Res 43: 4227
Bhuyan BK, Moy BC, Badiner GJ (1987) Characterization of B16 melanoma cells resistant to U71, 184 (a CC-1065 analog). Proc Am Assoc Cancer Res 28: 296
Buick R, Pullano R, Trent J (1985) Comparative properties of five ovarian adenocarcinoma cell lines. Cancer Res 45: 3668
Chabner BA, Myers CE (1989) Clinical pharmacology of cancer chemotherapy. In: Devita VT, Hellman S, Rosenberg SA (eds) Cancer principles and practice of oncology, 3rd edn. J. B. Lippincott, Philadelphia, p 369
Chou T, Talaly P (1984) Quantitative analysis of dose-effect relationships: the combined effects of multiple drugs or enzyme inhibitors. Adv Enzyme Regul 22: 27
Dekoning TF, Krueger WC, Wallace TL, Prairie MD, Warpehoski MA (1987) Influence of structure and chirality on the antitumor activity and biochemical effects of CC-1065 and its analogs. Proc Am Assoc Cancer Res 28: 262
Fogh J, Trempe G (1975) New human tumor cell lines. In: Fogh J (ed) Human tumor cells in vitro. Plenum Press, New York, p 115
Garewal H, Ahmann F, Schifman R, Celniker A (1986) ATP assay: ability to distinguish cytostatic from cytocidal anticancer drug effects. J Natl Cancer Inst 77: 1039
Geisinger K, Kute T, Pettenati M, Welander C, Dennard Y (1989) Characterization of a human ovarian carcinoma cell line with estrogen and progesterone receptors. Cancer 63: 280
Hanka LJ, Dietz A, Gerpheide SA, Kuentzel SL, Martin DG (1978) CC-1065 (NSC-298223), a new antitumor antibiotic. Production, in vitro biological activity, microbiological assay, and taxonomy of the producing organism. J Antibiot 31: 1211
Ichihashi H, Nogaki M, Yamauchi M, Tagaki H, Kondo T, Ozawa T (1986) A simple and rapid in vitro test for determining sensitivity of cancer cells to anticancer drugs. Jpn J Surg 16 (5): 380
Kingston RE, Sevin BU, Ramos R, Saks M, Donato D, Averette H (1988) Synergistic effects of cisplatin (CP) and cytosine arabinoside (CA) on ovarian cell lines demonstrated by dual parameter flow cytometry. Gynecol Oncol 32: 282
Kuzmits R, Rumpold H, Mueller M, Schoof G (1986) The use of bioluminescence to evaluate the influence of chemotherapeutic drugs on ATP-levels of malignant cell lines. J Clin Chem Clin Biochem 24: 293
Maehara Y, Anai H, Tamada R, Sugimachi K (1987) The ATP assay is more sensitive than succinate dehydrogenase inhibition test for predicting cell viability. Eur J Cancer Clin Oncol 23 (3): 273
Martin DG, Hanka LJ, Neil GL (1978) Isolation, characterization, and preliminary antitumor evaluation of CC-1065, a potent new agent from fermentation. Proc Am Assoc Cancer Res 19: 99
Martin DG, Chidester CG, Duchamp DJ, Mizask SA (1980) Structure of CC-1065 (NSC298223), a new antitumor antibiotic. J Antibiot 33: 902
Martin DG, Biles C, Gerpheide SA, Hanka LJ, Krueger WC (1981) CC-1065 (NSC298 223), a potent new antitumor agent. Improved production and isolation, characterization, and antitumor activity. J Antibiot 34: 1119
McGovren JP, Clarke GL, Pratt EA, Dekoning TF (1984) Preliminary toxicity studies with the DNA-binding antibiotic, CC-1065. J Antibiot 37: 63
Moyer JD, Henderson JF (1983) Ultrasensitive assay for ribonucleoside triphosphates in 50–1000 cells. Biochem Pharmacol 32: 3831
Nguyen HN, Sevin BU, Averette HE, Perras J, Ramos R, Penalver M, Donato D (1991) The effects of provera on chemotherapy of uterine cancer cell lines. Gynecol Oncol 42: 165
Nogaki M, Ichihashi H (1988) A new chemosensitivity test for cancer cells by measuring intracellular ATP content. Nippon Geka Gakkai Zasshi 89 (12): 1950
Petru E, Sevin BU, Petras J, Boike G, Ramos R, Nguyen H, Averette HE (1990) Comparative chemosensitivity profiles in four human ovarian carcinoma cell lines measuring ATP bioluminescence. Gynecol Oncol 38: 155
Reynolds VL, McGovren JP, Hurley LH (1986) The chemistry, mechanism of action and biological properties of CC-1065, a potent antitumor antibiotic. J Antibiot 39: 319
Saeki T, Jinushi K, Kim R, Kirihara Y, Takagami S, Nishiyama M, Saeki K, Niimoto M, Hattori T (1989) Comparison of the succinic dehydrogenase inhibition test with the adenosine triphosphate inhibition assay for human solid tumors as an in vitro chemosensitivity test. Gan To Kagaku Ryoho 16 (5): 2025
Selby P, Buick R, Tannock I (1983) A critical appraisal of the human tumor stem cell assay. N Engl J Med 308: 129–134
Sevin BU, Peng ZL, Perras JP, Ganjei P, Penalver M, Averette HE (1988) Application of an ATP-bioluminescence assay in human tumor chemosensitivity testing. Gynecol Oncol 31: 191
Swenson DH, Li LH, Hurley LH, Rokem JS, Petzold GL (1982) Mechanism of interaction of CC-1065 (NSC298223) with DNA. Cancer Res 42: 2821
Von Hoff DD, Casper J, Bradley E (1981) Association between human tumor colony-forming assay results and response of an individual patient's tumor to chemotherapy. Am J Med 70: 1027
Warpehoski MA (1989) CC-1065 analogs: therapeutic potential of a sequence-specific drug-DNA reaction. Proc Am Assoc Cancer Res 30: 668
Warpehoski MA, Gebhard I, Kelly RC, Krueger WC, Li LH (1988) Stereoelectronic factors influencing the biological activity and DNA interaction of synthetic antitumor agents modeled on CC-1065. J Med Chem 31: 590
Wierenga W, Bhuyan BK, Kelly RC, Krueger WC, Li LH (1986) Antitumor activity and biochemistry of novel analogs of the antibiotic CC-1065. Adv Enzyme Regul 25: 141
Author information
Authors and Affiliations
Additional information
Supported in part by an American Cancer Society Clinical Oncology Fellowship and an American Cancer Society/Florida Division Startup Grant (both awarded to H. N. N.)
Rights and permissions
About this article
Cite this article
Nguyen, H.N., Sevin, BU., Averette, H. et al. In vitro evaluation of a novel chemotherapeutic agent, adozelesin, in gynecologic-cancer cell lines. Cancer Chemother. Pharmacol. 30, 37–42 (1992). https://doi.org/10.1007/BF00686483
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00686483