Abstract
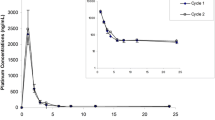
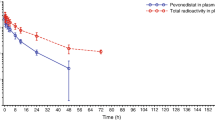
The pharmacokinetics and ex vivo pharmacodynamics studies oncis-malonato[(4R,5R)-4,5-bis (aminomethyl)-2-isopropyl-1,3-dioxolane]platinum(II) (SKI 2053R, NSC D644591), cisplatin (CDDP), and carboplatin (CBDCA) were performed in beagle dogs. Equitoxic doses of SKI 2053R, CDDP, and CBDCA (7.5, 2.5, and 15.0 mg/kg, respectively) were given by i.v. bolus to three beagle dogs in a randomized crossover study. Plasma samples were analyzed for platinum by flameless atomic absorption spectrophotometry. Plasma concentrations of total and ultrafiltrable platinum for the three drugs declined in a biexponential fashion. The mean area under the concentration-time curve (AUC0→∞) determined for ultrafiltrable platinum derived from SKI 2053R, as an active component, was 7.72±2.74 μg h ml−1 (mean ± SD), with an initial half-life of 0.37±0.20 h, a terminal half-life of 2.19±0.93 h, a total clearance of 16.83±4.76 ml min−1 kg−1, and a steady-state volume of distribution of 1.57±0.30 l/kg. The ex vivo antitumor activity of SKI 2053R was assessed using the ultrafiltrable plasma against two human lung-adenocarcinoma cell lines (PC-9 and PC-14) and five stomach-adenocarcinoma cell lines (MKN-45, KATO III, SNU-1, SNU-5, and SNU-16) by tetrazolium-dye (MTT) assay and was compared with that of CDDP and CBDCA using an antitumor index (ATI) determined from the ex vivo pharmacodynamic results of inhibition rates (%) versus time curves. The mean ATI value was shown to be ranked in the following order: SKI 2053R > CBDCA > CDDP. The mean ATI values recorded for SKI 2053R and CBDCA were significantly (P<0.05) higher than that noted for CDDP; however, no statistically significant difference was observed between SKI 2053R and CBDCA, suggesting that the antitumor activity of SKI 2053R is superior to that of CDDP and is equivalent to that of CBDCA. These results suggest that SKI 2053R is a promising candidate for further development as a clinically useful anticancer drug.
Similar content being viewed by others
References
Carter SK (1984) Cisplatin-past, present and future. In: Hacker MP, Douple EB, Krakoff IH (eds) Platinum coordination complexes in cancer chemotherapy. Martinus Nijhoff, Boston, p 359
Cole WC, Wolf W (1980) Preparation and metabolism of a cisplatin/serum protein complex. Chem Biol Interact 30: 223
Durant JR (1980) Cisplatin: a clinical overview. In: Prestayko AW, Crooke ST, Carter SK (eds) Cisplatin, current status and new developments. Academic Press, New York, p 317
Gore M, Fryatt I, Wiltshaw E, Dawson T, Robinson B, Calvert A (1989) Cisplatin/carboplatin cross-resistance in ovarian cancer. Br J Cancer 60: 767
Gormley PE, Bull JM, Leroy AF, Cysyk R (1979) Kinetics ofcis-dichlorodiammineplatinum. Clin Pharmacol Ther 25:351
Harrap KR (1985) Preclinical studies identifying carboplatin as a visible cisplatin alternative. Cancer Treat Rev 12 [Suppl A]: 21
Kang K-S, Shin D-J, Cho J-J, Kim H-O, Kim B-H, Lee Y-S (1992) Acute toxicity ofcis-malonato[(4R,5R)-4,5-bis(aminomethyl)-2-isopropyl-1,3-dioxolane]platinum(II) (SKI 2053R) in rats and mice. Korean J Toxicol 8: 205
Kim D-K, Kim G, Gam J, Cho Y-B, Kim H-T, Tai J-H, Kim KH, Hong W-S, Park J-G (1994) Synthesis and antitumor activity of a series of [2-substituted-4,5-bis(aminomethyl)-1,3-dioxolane]platinum(II) complexes. J Med Chem 37: 1471
Kim D-K, Kim H-T, Cho Y-B, Tai JH, Ahn JS, Kim T-S, Kim KH, Hong W-S (1995) Antitumor activity ofcis-malonato [(4R, 5R)-4,5-bis(aminomethyl)-2-isopropyl-1,3-dioxolane] platinum(II), a new platinum analogue, as an anticancer agent. Cancer Chemother Pharmacol 35: 441
Kim H-O, Kang K-S, Shin D-J, Cho J-J, Kim B-H, Seo K-W, Nam K-H, Lee Y-S (1992) Subacute toxicity ofcis-malonato [(4R,5R)-4,5-bis(aminomethyl)-2-isopropyl-1,3-dioxolane]-platinum(II) (SKI 2053R) in rats. Korean J Toxicol 8: 217
Kracker AJ, Hoeschele JD, Elliott WL, Showalter HDH, Sercel AD (1992) Anticancer activity in murine and human tumor cell lines of bis(platinum) complexes incorporating straight-chain aliphatic diamine linker groups. J Med Chem 35: 4526
Lee Y-S, Kang K-S, Shin D-J, Cho J-J, Kim H-O, Kim B-H, Lim Y-K (1992) Subacute toxicity ofcis-malonato[(4R,5R)-4, 5-bis(aminomethyl)-2-isopropyl-1,3-dioxolane]platinum(II) (SKI 2053R) in beagle dogs. Korean J Toxicol 8: 235
Loeher PJ, Einhorn LH (1984) Cisplatin. Ann Intern Med 100: 704
Mellish KJ, Kelland LR, Harrap KR (1993) In vitro platinum drug chemosensitivity of human cervical squamous cell carcinoma cell lines with intrinsic and acquired resistance to cisplatin. Br J Cancer 68: 240
Ozols RF, Corden BJ, Jacob J, Wesley MN, Ostechega Y, Young RC (1984) High-dose cisplatin in hypertonic saline. Ann Intern Med 100: 19
Ozols RF, Ostechega Y, Myers CE, Young RC (1985) High-dose cisplatin in hypertonic saline in refractory ovarian cancer. J Clin Oncol 3: 1246
Park J-G, Frucht H, LaRocca RV, Bliss DP Jr, Kurita Y, Chen T-R, Henslee JG, Trepel JB, Jensen RT, Johnson BE, Bang Y-J, Kim J-P, Gazdar AF (1990) Characteristics of cell lines established from human gastric carcinoma. Cancer Res 50: 2773
Rose WC, Schurig JE (1985) Preclinical antitumor and toxicologic profile of carboplatin. Cancer Treat Rev 12 [Suppl A]: 1
Sasaki Y, Shinkai T, Eguchi K, Tamura T, Ohe Y, Ohmori T, Saijo N (1991) Prediction of the antitumor activity of new platinum analogs based on their ex vivo pharmacodynamics as determined by bioassay. Cancer Chemother Pharmacol 27: 263
Vijgh WJF van der (1991) Clinical pharmacokinetics of carboplatin. Clin Pharmacokinet 4: 242
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Kim, DK., Kim, HT., Tai, J.H. et al. Pharmacokinetics and antitumor activity of a new platinum compound,cis-malonato[(4R,5R)-4,5-bis(aminomethyl)-2-isopropyl- 1, 3-dioxolane]platinum(II), as determined by ex vivo pharmacodynamics. Cancer Chemother. Pharmacol. 37, 1–6 (1995). https://doi.org/10.1007/BF00685622
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00685622