Abstract
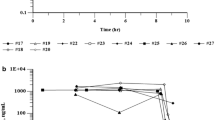
A total of 21 patients with advanced cancer were entered into a phase I study to determine the maximum tolerable dose (MTD) of liposome-encapsulated doxorubicin (LED) given weekly for 3 consecutive weeks at doses of 20, 30, or 37.5 mg/m2 per week. For a comparison of the pharmacokinetic behavior of LED with that of standard-formulation doxorubicin, 13 patients received a dose of standard-formulation doxorubicin 2 weeks prior to the first dose of LED. All doses were given by 1-h infusion through a central vein. Toxicity was evaluated in 22 courses delivered to 17 patients. The MTD with this schedule was 30 mg/m2 per week×3. The single patient treated at 37.5 mg/m2 weekly could not complete the entire course due to myelosuppression. At the dose of 30 mg/m2 per week, three of eight patients had grade ≥3 leukopenia. Other toxicities included mild to moderate thrombocytopenia, nausea, vomiting, fever, alopecia, diarrhea, fatigue, stomatitis, and infection. At the dose of 30 mg/m2 per week, the total doxorubicin AUC and peak total doxorubicin concentrations in plasma were 8.75±8.80 μM h (mean±SD) and 3.07±1.45 μM, respectively, after LED administration. The total doxorubicin AUC and peak total doxorubicin concentrations in plasma were 3.92±2.47 μM h and 2.75±2.70 μM, respectively, after the infusion of standard-formulation doxorubicin. The total body clearance of doxorubicin was 18.42±11.23 l/h after the infusion of LED and 31.21±15.48 l/h after the infusion of standard-formulation doxorubicin. The mean elimination half-lives of doxorubicin were similar: 8.65±5.16 h for LED and 7.46±5.16 h for standard-formulation doxorubicin. Interpatient variability in pharmacokinetic parameters as demonstrated by the percentage of coefficients of variation was 33%–105%. There was no relationship between the percentage of WBC decrease or the duration of WBC suppression and the total doxorubicin or doxorubicinol AUC. There was no correlation between the duration of leukopenia and drug exposure as reflected by the AUC of liposome-associated doxorubicin. LED can be given in doses similar to those of standard-formulation doxorubicin and produces acute toxicities similar to those caused by standard doxorubicin.
Similar content being viewed by others
Abbreviations
- MTD:
-
maximum tolerable dose
- LED:
-
liposome-encapsulated doxorubicin
- AUC:
-
area under the plasma concentration x time curve
- WBC:
-
white blood cell count
- PLT:
-
platelet count
- ECOG:
-
Eastern Cooperative Oncology Group
- EKG:
-
electrocardiogram
- MUGA:
-
multigated nuclide scan
- CLTB:
-
total body clearance
- PC:
-
phosphatidylcholine: PG, phosphatidylglycerol
- PEG-DSPE:
-
polyethylene glycol conjugated to distearoyl phosphatidylethanolamine
- HSPC:
-
hydrogenated soy phosphatidylcholine
- chol:
-
cholesterol
References
Andrews PA, Brenner DE, Chou FE, Kubo H, Bachur NR (1980) Facile and definitive determination of human Adriamycin and daunorubicin metabolites by high-pressure liquid chromatography. Drug Metab Dispos 8: 152
Balazsovitz JAE, Mayer LD, Bally MB, Cullis PR, McDonell M, Ginsberg RS, Falk RE (1989) Analysis of the effect of liposome encapsulation on the resistant properties, acute and cardiac toxicities, and antitumor efficacy of doxorubicin. Cancer Chemother Pharmacol 23: 81
Bally MB, Nayar R, Masin D, Cullis PR, Mayer LD (1990) Studies on the myelosuppressive activity of doxorubicin entrapped in liposomes. Cancer Chemother Pharmacol 27: 13
Gabizon A (1992) Selective tumor localization and improved therapeutic index of anthracyclines encapsulated in long-circulating liposomes. Cancer Res 52: 891
Gabizon A, Goren D, Fuks Z, Meshorer A, Berenholz Y (1985) Superior therapeutic activity of liposome-associated Adriamycin in a murine metastatic tumor model. Br J Cancer 51: 681
Gabizon A, Peretz T, Sulkes A, Amselem A, Ben-Yosef R, Ben-Baruch N, Catane R, Biran S, Barenholz Y (1989) Systemic administration of doxorubicin-containing liposomes in cancer patients: a phase I study. Eur J Cancer Clin Oncol 25: 1795
Gabizon A, Shiota R, Papahadjopoulos D (1989) Pharmacokinetics and tissue distribution of doxorubicin encapsulated in stable liposomes with long circulation times. J Natl Cancer Inst 81: 1484
Gabizon A, Chisin R, Amselem S, Druckmann S, Cohen R, Goren D, Fromer I, Peretz T, Sulkes A, Barenholz Y (1991) Pharmacokinetic and imaging studies in patients receiving a formulation of liposome-associated Adriamycin. Br J Cancer 64: 1125
Gregoriadis G (1976) The carrier potential of liposomes in biology and medicine. New Engl J Med 295: 705
Herman EH, Rahman A, Ferrans VJ, Vick JA, Schein PS (1983) Prevention of chronic doxorubicin cardiotoxicity in beagles by liposomal encapsulation. Cancer Res 43: 5427
Knott GD (1979) MLAB: a mathematical modeling tool. Comput Programs Biomed 10: 271
Mace K, Mayhew E, Mihich E, Ehrke MJ (1989) Alterations in murine host defense functions by Adriamycin or liposome-encapsulated Adriamycin. Cancer Res 48: 130
Mayer LD, Bally MB, Hope MJ, Cullis PR (1985) Uptake of antineoplastic agents into large unilamellar vesicles in response to a membrane potential. Biochim Biophys Acta 816: 294
Oudard S, Thierry A, Jorgensen TJ, Rahman A (1991) Sensitization of multidrug-resistant colon cancer cells to doxorubicin encapsulated in liposomes. Cancer Chemother Pharmacol 28: 259
Papahadjopoulos D, Allen TM, Gabizon A, Mayhew E, Matthay K, Huang SK, Lee KD, Woodle MC, Lasic DD, Redemann C, Martin FJ (1991) Sterically stabilized liposomes: improvements in pharmacokinetics and antitumor therapeutic efficacy. Proc Natl Acad Sci USA 88: 11460
Perez-Soler R (1989) Liposomes as carriers of antitumor agents: toward a clinical reality. Cancer Treat Rev 16: 67
Rahman A, Treat J, Roh J-K, Potkul LA, Alvord WG, Forst D, Woolley PV (1990) A phase I clinical trial and pharmacokinetic evaluation of liposome-encapsulated doxorubicin. J Clin Oncol 8: 1093
Storm G, Hoesel QGCM van, Groot G de, Kep W, Steerenberg PA, Hillen FC (1989) A comparative study on the antitumor effect, cardiotoxocity and nephrotoxicity of doxorubicin given as a bolus, continuous infusion and entrapped in liposomes in the Lou/M Wsl rat. Cancer Chemother Pharmacol 24: 341
Thierry AR, Jorgensen TJ, Forst D, Belli JA, Drilschilo A, Rahman A (1989) Modulation of multidrug resistance in Chinese hamster cells by liposome-encapsulated doxorubicin. Cancer Commun 1: 311
Thies RL, Cowens DW, Cullis PR, Bally MB, Mayer LD (1990) Method for rapid separation of liposome-associated doxorubicin from free doxorubicin in plasma. Anal Biochem 188: 65
Treat J, Greenspan A, Forst D, Sanchez JA, Ferrans VJ, Potkul LA, Woolley PV, Rahman A (1990) Antitumor activity of liposome-encapsulated doxorubicin in advanced breast cancer: phase II study. J Natl Cancer Inst 82: 1706
Author information
Authors and Affiliations
Additional information
This work was supported by DHHS, NCl NO-l-CM 07 303 and by a Career Development Award from the American Cancer Society (to B. A. C.)
Rights and permissions
About this article
Cite this article
Conley, B.A., Egorin, M.J., Whitacre, M.Y. et al. Phase I and pharmacokinetic trial of liposome-encapsulated doxorubicin. Cancer Chemother. Pharmacol. 33, 107–112 (1993). https://doi.org/10.1007/BF00685327
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00685327