Summary
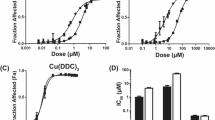
The antitumor activity of eight new metal complexes (three platinum, one titanium, four ruthenium derivatives) was investigated in a cisplatin (DDP)-sensitive (O-342) and a DDP-resistant (O-342/DDP) ovarian tumor line using the bilayer soft-agar assay. A continuous exposure set up at logarithmically spaced concentrations was used to test the drugs; to uncover possible pharmacokinetic features, a short-term exposure was additionally included for selected compounds. DDP served as the reference drug. The following compounds were investigated: 18-crown-6-tetracarboxybis-diammineplatinum(II) (CTDP),cis-aminotrismethylenephosphonato-diammineplatinum(II) (ADP),cis-diamminecyclohexano-aminotrismethylenephosphonato-platinum(II) (DAP), diethoxybis(1-phenylbutane-1,3-dionato)titanium(IV) (DBT, budotitane),trans-imidazolium-bisimidazoletetrachlororuthenate(III) (ICR),trans-indazolium-tetrachlorobisindazoleruthenate(III) (IndCR),cis-triazolium-tetrachlorobistriazoleruthenate(III) (TCR) andtrans-pyrazolium-tetrachlorobispyrazoleruthenate(III) (PCR). Of the new metal complexes, CTDP was the most active compound in O-342, resulting in a percentage of control plating efficiency (±SE) of 1±1, 12±8 and 40±21 following continuous exposure to 10, 1 and 0.1 μm, respectively, and was thus comparable to DDP at equimolar concentrations. In the resistant line, 10 μm CTDP reduced colony growth to 18%±8%, whereas an equimolar concentration of DDP effected a reduction to 26%±9%. During short-term exposure, CTDP was inferior to DDP, which may be ascribed to the stability of the bis-dicarboxylate platinum ring system. The titanium compound DBT, in contrast, showed promising effects at its highest concentration (100 μm) during short-term exposure in both lines; at this concentration the activity in O-342/DDP was higher than that in O-342 (7%±7% vs 34%±17% of control plating efficiency at 100 μm). All ruthenium complexes showed higher activity in the resistant line O-342/DDP than in the sensitive counterpart. ICR was the most active compound. Following continuous exposure of O-342/DDP cells to 10 μm ICR, colony growth was reduced to 18%±4% that of controls. Further studies should concentrate on CTDP and ICR for the following reasons: the activity of CTDP was equal to that of DDP at equimolar concentrations during continuous exposure; considering that the in vivo toxicity of DDP was 3-fold that of CTDP, an increase in the therapeutic index of CTDP would be expected. ICR showed the best effect of all ruthenium complexes; it was superior to DDP in the resistant line.
Similar content being viewed by others
References
Alberts DS, Salmon SE, Chen HSG, Moon TE, Young L, Surwit EA (1981) Pharmacologic studies of anticancer drugs with the human tumor stem cell assay. Cancer Chemother Pharmacol 6:253–264
Alessio E, Attia W, Calligaris M, Cauci S, Dolzani L, Mestroni C, Monti-Bragadin G, Quadrifoglio F, Sava G, Tamaro M, Zorzet S (1988) Metal complexes of platinum group: the promising antitumor features ofcis-dichlorotetrakis (dimethylsulfoxide) Ru(II), [cis-RuCl2(Me2SO)4] and related complexes. In: Nicolini M (ed) Platinum and other metal coordination compounds in cancer chemotherapy. Martinus Nijhoff, Boston, pp 617–633
Alley MC, Uhl CB, Lieber MM (1982) Improved detection of drug cytotoxicity in the soft agar colony formation assay through use of a metabolizable tetrazolium salt. Life Sci 31:3071–3078
Berger MR, Garzon FT, Keppler BK, Schmähl D (1989) Efficacy of new ruthenium complexes against chemically-induced autochthonous colorectal carcinoma in rats. Anticancer Res 9:761–765
Bischoff H, Berger MR, Keppler BK, Schmähl D (1987) Efficacy of beta-diketonato complexes of titanium, zirconium and hafnium against chemically induced autochthonous colonic tumors in rats. J Cancer Res Clin Oncol 113:446–450
Burchenal JH, Irani G, Kern K, Lokys L, Turkevic J (1980) 1,2-Diaminocyclohexane platinum derivatives of potential clinical value. Recent Results Cancer Res 74:146–155
Calvert AH, Harland SJ, Newell DR, Siddik H, Jones AC, McElwain TJ, Raju S, Wiltshaw E, Smith IE, Baker JM, Peckham MJ, Harrap KR (1982) Early clinical studies withcis-diammine-1,1-cyclobutane-dicarboxylate platinum II. Cancer Chemother Pharmacol 9:140–147
Chen G, Frei E, Zeller WJ (1989) Determination of intracellular reduced glutathione and glutathione related enzyme activities in cis-platin-sensitive and-resistant experimental ovarian carcinoma cell lines. Cancer Lett 46:207–211
Clarke MJ, Galang RD, Rodriguez VM, Kumar R, Pell S, Bryan DM (1988) Chemical considerations in the design of ruthenium anticancer agents: In: Nicolini M (ed) Platinum and other metal coordination compounds in cancer chemotherapy. Martinus Nijhoff, Boston, pp 582–600
Cleare MJ, Hydes PC, Malerbi BW, Watkins DM (1978) Antitumor platinum complexes: relationships between chemical properties and activity. Biochimie 60:835–860
Fiebig HH, Schmid JR, Bieser W, Henss H, Lohr GW (1987) Colony assay with human tumor xenografts, murine tumors and human bone marrow. Potential for anticancer drug development. Eur J Cancer Clin Oncol 23:937–948
Garzon FT, Berger MR, Keppler BK, Schmähl D (1987) Comparative antitumor activity of ruthenium derivatives with 5′-deoxy-5-fluorouridine in chemically induced colorectal tumors in SD rats. Cancer Chemother Pharmacol 19:347–349
Graeff A de, Slebos RJC, Rodenhuis S (1988) Resistance to cisplatin and analogues: mechanisms and potential clinical implications. Cancer Chemother Pharmacol 22:325–332
Hamburger AW, Salmon SE (1977) Primary bioassay of human tumor stem cells. Science 197:461–463
Harrap KR, Jones M, Wilkinson CR, Clink HM, Sparrow S, Mitchley BCV, Clarke S, Veasey A (1980) Antitumor, toxic, and biochemical properties of cisplatin and eight other platinum complexes. In: Prestayko AW, Crooke ST, Carter SK (eds) Cisplatin — Current status and new developments. Academic Press, New York, pp 193–212
Holland JF, Bruckner HW, Cohen CJ, Wallach RC, Gusberg SB, Greenspan EM, Goldberg J (1980) Cisplatin therapy of ovarian cancer. In: Prestayko AW, Crooke ST, Carter SK (eds) Cisplatin — current status and new developments. Academic Press, New York, pp 383–392
Hrubisko M, Balazova E, Ujhazy V (1984) Drug resistance induction and cross-resistance studies with Pt-complexes. Neoplasma 31:649–653
Kaufmann M, Buse KD, Kubli F (1984) Der Softagar-Tumorzellkolonie-Assay zur In-vitro-Chemoresistenztestung und als Prognosefaktor bei menschlichen Tumoren. In: Jonat W, Kaufmann M, Kubli F, Maass H (eds) Prätherapeutische Tumortestung (Aktuelle Onkologie 16). Karger, Munich, pp 132–136
Keller HJ, Keppler B, Schmähl D (1982) Antitumor activity ofcis-dihalogeno-bis-(1-phenyl-1,3-butanedionato)titanium(IV) compounds against Walker 256 carcinosarcoma — a new class of antineoplastic agents. Arzneimittelforschung/Drug Res 32:806–807
Keppler BK, Heim ME (1988) Antitumor-active bis-beta-diketonato metal complexes: budotitane — a new anticancer agent. Drugs Fut 13:637–652
Keppler BK, Rupp W (1986) Antitumor activity of imidazoliumbisimidazole-tetrachlororuthenate(III). J Cancer Res Clin Oncol 111:166–168
Keppler BK, Rupp W, Juhl UM, Endres H, Niebl R, Balzer W (1987) Synthesis, molecular structure and tumor-inhibiting properties oftrans-bis(imidazole)tetrachlororuthenate(III) and its methyl-substituted derivatives. Inorg Chem 26:4366–4370
Keppler BK, Henn M, Juhl UM, Berger MR, Niebl R, Wagner FE (1989) New ruthenium complexes for the treatment of cancer. Prog Clin Biochem Med 10:41–69
Klenner T, Keppler BK, Amelung F, Schmähl D (1989) Aminotris-(methylenephosphonato)-diamminoplatinum II (ADP), a new anticancer agent superior to cisplatin (CDDP) in the transplantable rat osteosarcoma. J Cancer Res Clin Oncol 115 [Suppl]:S 54
Köpf-Maier P, Moormann A, Köpf H (1985) Activity of titanocene dihalides against a human colon carcinoma heterotransplanted to athymic mice. Eur J Cancer Clin Oncol 21:853–857
Shoemaker RH, Wolpert-DeFilippes MK, Kern DH, Lieber MM, Makuch RW, Melnick NR, Miller WT, Salmon SE, Simon RM, Venditti JM, VonHoff D (1985) Application of a human tumor colony-forming assay to new drug screening. Cancer Res 45:2145–2153
VonHoff DD, Clark GM, Stogdill BJ, Sarosdy MF, O'Brien MT, Casper JT, Mattox DE, Page CP, Cruz AB, Sandbach JF (1983) Prospective clinical trial of a human tumor cloning system. Cancer Res 43:1926–1931
Zeller WJ, Chen G (1990) Chemosensitization of experimental ovarian tumors to cisplatin (DDP) byd,l-buthionine (S,R)-sulfoximine (BSO) or 3-amino-benzamide (3-AB) in vivo. Proc Am Assoc Cancer Res 31:406
Zeller WJ, Frühauf S, Chen G, Keppler BK, Frei E, Kaufmann M (1991) Chemoresistance in rat ovarian tumours Eur J Cancer, in press
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Frühauf, S., Zeller, W.J. In vitro evaluation of platinum, titanium and ruthenium metal complexes in cisplatin-sensitive and-resistant rat ovarian tumors. Cancer Chemother. Pharmacol. 27, 301–307 (1991). https://doi.org/10.1007/BF00685116
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00685116