Abstract
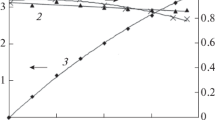
An electrochemical device for oxygen production which makes oxygen on both cathode and anode from air has been developed. The electrolytic cell consists of an air cathode, a catalytic decomposition mesh and a nickel anode. The electrolyser is composed of six single cells. The working surface area of a single cell is 0.02 m2. Performance depends on factors such as the basic electrolyte concentration, temperature, air flow rate, gas flow distribution and current density. When the device is operated with 7m KOH at room temperature and with an air flow rate of 0.4m3 h−1 and current of 20 A, the oxygen evolution rate is 0.036m3 h−1 and the oxygen purity is 99.5%. The current efficiency is 72.0%. The device has operated continuously for 12 months with stable performance.
Similar content being viewed by others
References
R. A. Wynveen and K. M. Montgomery,J. Electrochem. Soc. 114 (1967) 589.
A. C. C. Tseung and S. M. Jasem,J. Appl. Electrochem. 11 (1981) 209–15.
Nagakazu Furuya, Masanori Kanazawa and Satoshi Motoo,Denki Kagaku 55(12) (1987) 944–45.
C. Olomon and A. P. Watkinson,J. Appl. Electrochem. 9 (1979) 117–23.
Idem,Can. J. Chem. Eng. 53 (1975) 268–73;54 (1976) 31218.
M. Sudoh, H. Kitaguchi and K. Koide,J. Chem. Eng. Japan 18(5) (1985) 409–14;18(4) (1985) 364–71.
C. A. Ma, Y. Zhuwang, Z. Yuanhong and C. Chuanxin,Phys. Chem. Acta, China 6(5) (1990) 622–28.
C. A. Ma,Chinese Patent ZL92112233. 0 (1992)
A. Feihu and L. Guoliang, ‘Study of the Preparation and Application of the Oxygen Cathode’, Zhejiang University of Technology Data (1993).
C. A. Ma and W. G. Yu, ‘Study of Electrochemical Medical Oxygen-making Machine (prototype authentication)’, Zhejiang University of Technology Data (1995) pp. 1–60.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ma, C.A., Yu, W.G. An electrochemical device for oxygen production avoiding the generation of hydrogen. J Appl Electrochem 26, 881–885 (1996). https://doi.org/10.1007/BF00683751
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00683751