Abstract
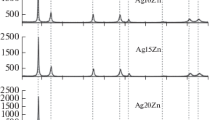
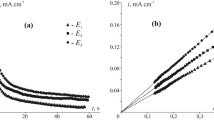
Internal oxidation of dilute silver alloys containing Al, Mg, Zn, Cu, and Sn was studied in air at temperatures between 573 K and 1173 K. Electrical resistivity, gravimetric, and gas-extraction measurements were made. The general trend of the resistivity is that it increases upon oxidation at lower temperatures and the resistivity decreases at higher temperatures in all of these alloys except Ag-Mg, in which it increases even at 1173 K. The increase in resistivity is considered to be related to the formation of clusters having excess oxygen. A detailed investigation was performed on Ag-Al alloys. The O/Al ratio in the clusters in Ag-2.2 at.% Al is much higher on oxidation at 773 K than for stoichiometric Al2O3 at 1173 K. The clusters release the excess oxygen on subsequent annealing at high temperatures, and decompose to stable Al2O3 at 1173 K.
Similar content being viewed by others
References
A. Combe and J. Cabane,Oxid. Met. 21, 21 (1984).
M. J. Klein and R. A. Huggins,Trans. Am. Soc. Met. 55, 259 (1962).
A. Combe, L. Charrin, G. Moya, and J. Cabane,Acta Metall. 31, 1019 (1983).
F. R. Fickett,Mater. Sci. Eng. 14, 199 (1974).
N. Yoshida, Y. Tomii, J. Takada, S. Kikuchi, and M. Koiwa,Trans. Jpn. Inst. Met. 29, 693 (1988).
R. Lacroix,Mem. Scient. Rev. Met. 63, 693 (1966).
L. S. Darken,Trans. Am. Soc. Met. 54, 600 (1961).
E. Thilo and W. Gessner,Zeit. Anorg. All. Chem. 345, 151 (1966).
J. S. Hirschhorn and F. V. Lenel,Trans. Am. Soc. Met. 59, 208 (1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Takada, J., Tomii, Y., Yoshida, N. et al. Internal oxidation of dilute silver alloys. Oxid Met 37, 13–22 (1992). https://doi.org/10.1007/BF00665628
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00665628