Abstract
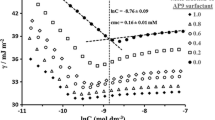
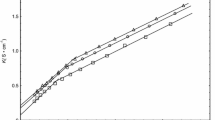
The enthalpy-entropy compensation in ionic surfactant micellization process over a large temperature range is examined. The surfactants SDS and C16TAB are investigated experimentally, and the enthalpy and entropy changes are evaluated based on phase separation or mass action models together with the other three surfactant systems. The relationship between compensation temperature and the reference temperatures is discussed.
Similar content being viewed by others
Abbreviations
- ΔC p :
-
heat capacity change, J/mol-K
- CMC:
-
critical micelle concentration,M
- CMC0 :
-
critical micelle concentration atT=T 0,M
- ΔG :
-
Gibbs free energy change, kJ/mol
- ΔH :
-
enthalpy chang, kJ/mol
- Δh c :
-
enthalpy change for transfer of a methylene group to water, kJ/mol
- R :
-
gas constant, 8.314 J/mol-K
- ΔS :
-
entropy change, J/mol-K
- ΔS c :
-
entropy change for transfer of a methylene group to water, J/mol-K
- ΔS * :
-
entropy change atT=T *, J/mol-K
- T :
-
temperature,K
- T c :
-
compensation temperature, K
- T H :
-
temperature at which ΔH=0, K
- T 0 :
-
temperature at the minimum point, K
- T * :
-
112°C
- β:
-
degree of dissociation
References
Tanford C (1980) The Hydrophobic Effect. 2nd Edn., John-Willy, NY
Hunter RJ (1989) Foundations of Colloid Science, Vol I, Oxford press, NY
LaMesa C (1990) J Phys Chem 94:323–326
Frank HS, Evans MW (1954) J Chem Phys 13:507–532
Shinoda K, Kobayashi, M, Yamaguchi N (1987) J Phys Chem 91:5292–5294
Baldwins RL (1986) Proc Natl Acad Sci USA 83:8069–8072
Muller N (1988) J Soln Chem 17:661–672
Muller N (1990) Acc Chem Re 23:23–28
Murphy KP, Gill SJ (1989) Pure Appl Chem 61:1097–1104
Murphy KP, Privalov PL, Gill SJ (1990) Science 247:559–561
Gilli P, Ferretti V, Gilli G, Borea PA (1993) J Phys Chem 98:1515–1518
Madan B, Lee B (1994) Biophys Chem 51:279–289
Makhatadze GI, Privalov PL (1994) Biophys Chem 51:291–309
Murphy KP (1994) Biophys Chem 51:311–326
Jolicoeur, Philip PR (1974) Can J Chem 52:1834–1839
Goto A, Takemoto M, Endo F (1985) Bull Chem Soc Jpn 58:247–251
Bedo Zs, Berecz E, Lakatos I (1992) Colloid Polym Sci 270:799–805
Krishnan CV, Friedman HL (1973) J Soln Chem 2:37
Krishnan CV, Friedman HL (1973) J Soln Chem 2:119
Evans DF, Wightman PJ (1982) J Colloid Interf Sci 86:515–524
Nusselder JJH, Engberts JBFN (1992) J Colloid Interf Sci 148:353–361
Hamann SD (1978) Aus J Chem 31:919–921
Holtzer A, Holtzer MF (1974) J Phys Chem 78:1442–1443
Lee DJ (1993) Bull Coll Eng, NTU, 58:1–10
Causi S, De Lisi R, Milioto S, Tirone N (1991) J Phys Chem 95:5664–5673
Wennerstrom H, Lindman B (1979) Phys Rep 52:1–86
Gill SJ, Nichols NF, Wadso I (1976) J Chem Thermodyn 8:445
Naghibi H, Dec SF, Gill SJ (1986) J Phys Chem 90:4621; Naghibi H, Gill SJ (1987) J Phys Chem 91:245; Naghibi H, Ownby DW, Gill SJ (1987) J Chem Eng Data 32:422
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lee, D.J. Enthalpy-entropy compensation in ionic micelle formation. Colloid Polym Sci 273, 539–543 (1995). https://doi.org/10.1007/BF00658683
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00658683