Abstract
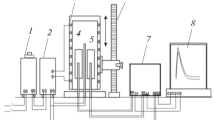
An investigation was carried out on the sulfidation kinetics of manganese plates by thermogravimetry in pure sulfur vapor at low pressures, 4.5×10−5< pS2 (atm)<7.2×10−4, at temperatures between 973 and 1173 K. The reaction kinetics were parabolic and the sulfidation rate constant at 1073 K was proportional to\(p_{S_2 }^{{1 \mathord{\left/ {\vphantom {1 n}} \right. \kern-\nulldelimiterspace} n}} \) where n = 6.4±0.8. The activation energy for the sulfidation reaction at\(p_{S_2 } = 8.1x10^{ - 5} \) atm was 25,000±3000 cal·mol−1. The α-MnS developed a preferred orientation texture, and grain growth was observed even though the metal exhibited no preferred orientation.
Similar content being viewed by others
References
K. Nishida, T. Narita, T. Tani, and G. Sasaki,Oxid. Met. 14, 1 (1980).
K. Ohta, K. Fueki, and T. Mukaibo,Denki Kagaku 38, 11 (1970).
F. A. Elrefaie and W. W. Smeltzer,Oxid. Met. 16, 267 (1981).
M. Lambertin, Thesis, Dijon, 1975.
F. A. Kröger,The Chemistry of Imperfect Crystals (North-Holland, New York, 1974).
G. Betrand and J. M. Chaix, Astérisque,Rev. Soc. Franç. Math. 98–99, 15 (1982).
C. Wagner, Diffusion and high temperature oxidation of metals, inAtom Movement (ASM, Cleveland, 1951), p. 153.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Perez, M., Larpin, J.P. Sulfidation of manganese in sulfur vapor at low pressure and temperatures between 873 and 1273 K. Oxid Met 21, 299–307 (1984). https://doi.org/10.1007/BF00656838
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00656838