Abstract
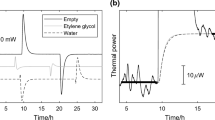
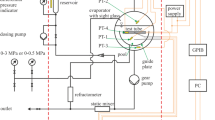
A flow method is described for the simultaneous measurement of volumetric specific heat capacities c p v and thermal expansion coefficients α of liquids, using a Picker heat capacity microcalorimeter. the method involves a simple sequence of operations in which the calorimeter unit is used alternately to measure a difference in volumetric specific heats at constant flow rate and temperature, or a flow rate variation resulting from a temperature scan on a flow cell. The precision of the method in α measurements is close to 1% which is comparable to that of current dilatometric methods; the overall time for the combined c p v-α measurement is less than 30 minutes. Typical uses and reliability of the method are illustrated through results for aqueous NaCl solutions, t-BuOH-water mixtures, and α,ω-diols (C2−C5) at 25°C. The α and c p v values in homologous series of normal alkanes, alcohols and diols appear well represented by equations comprising two contributions, one related to the hydrocarbon chain length, the other dependent on the number density of −OH groups. The general trends in these data and comparison with similar results for H2O2 and H2O illustrate the magnitude of the c p v and α ‘anomalies’ in liquid water.
Similar content being viewed by others
References
C. Jolicoeur, ‘Thermodynamic Flow Methods in Biochemistry, Calorimetry and Dilatometry,’ inMethods of Biochemical Analysis, D. Glick, ed. (John Wiley, New York, 1981), P. 171.
L. Ter Minassian, P. Pruzan, and A. Soulard,J. Chem. Phys. 75, 3064 (1981).
J. L. Fortier, M.-A. Simard, P. Picker, and C. Jolicoeur,Rev. Sci. Instrum. 50, 1474 (1979).
P. Picker, P.-A. Leduc, P. R. Philip, and J. E. Desnoyers,J. Chem. Thermodyn. 3, 631 (1971).
For recent references, see for example: G. Perron, A. Roux, and J. E. Desnoyers,Can. J. Chem. 59, 3049 (1981); J.-J. Spitzer, I. V. Olofsson, P. P. Singh, and L. G. Hepler,J. Chem. Thermodyn. 11, 233 (1979); F. J. Millero, G. Perron, and J. E. Desnoyers,J. Geophys. Chem. 78, 4499 (1973); A. L. Surdo, W.-Y. Wen, and C. Jolicoeur,J. Solution Chem. 8, 449 (1979); M. A. Willamanan, C. Casanova, A. H. Roux, and J.-P. E. Grolier,J. Chem. Thermodyn. 14, 251 (1982); D. Smith-Magowan, R. H. Wood, and D. M. Tillett,J. Chem. Eng. Data 27, 335 (1982); J. L. Fortier and G. C. Benson,J. Chem. Thermodyn. 8, 411 (1976).
J. L. Fortier, G. C. Benson, and P. Picker,J. Chem. Thermodyn. 8, 289 (1976).
P. Picker, E. Tremblay, and C. Jolicoeur,J. Solution Chem. 3, 377 (1974).
I. V. Olofsson,J. Chem. Thermodyn. 11, 1005 (1979).
J. E. Desnoyers, C. de Visser, G. Perron and P. Picker,J. Solution Chem. 5, 605 (1976).
M.-A. Simard and J. L. Fortier,Can. J. Chem. 59, 3208 (1981).
G. S. Kell,J. Chem. Eng. Data 20, 97 (1975).
N. S. Osborne, H. F. Stimson, and D. C. Ginnings,J. Res. Nat. Bur. Stand. 23, 197 (1939).
L. A. Dunn,Trans. Faraday Soc. 64, 2951 (1968).
F. T. Gucker,J. Am. Chem. Soc. 56, 1017 (1934).
F. J. Millero,J. Phys. Chem. 74, 356 (1970).
G. Perron, J. L. Fortier, and J. E. Desnoyers,J. Chem. Thermodyn. 7, 1177 (1975).
N. Desrosiers, Ph.D. Thesis, University of Sherbrooke (1975).
C. de Visser, G. Perron, and J. E. Desnoyers,Can. J. Chem. 55, 856 (1977).
A. Hvidt, R. Moss, and G. Nieslen,Acta Chem. Scand. 32, 274 (1978).
L. Avedikian, G. Perron, and J. E. Desnoyers,J. Solution Chem. 4, 331 (1975).
R. Arnaud, L. Avedikian, and J. P. Morel,J. Chem. Phys. 69, 45 (1972).
F. Franks and H. T. Smith,J. Chem. Eng. Data 13, 538 (1968).
C. Jolicoeur and G. Lacroix,Can. J. Chem. 54, 624 (1976).
E. M. Woolley and L. G. Hepler,Can. J. Chem. 55, 158 (1977).
C. Jolicoeur, L.-L. Lemelin, and R. Lapalme,J. Phys. Chem. 83, 2806 (1979).
R. C. Wilholt and B. J. Zwolinski,Journal of Physical and Chemical Reference Data Vol. 2, Suppl. No. 1 (1973).
TRC Tables, Selected Values of Properties of Chemical Compounds, Vol. I (d), Thermodynamics Research Center Data Project, Texas A&M University (1966).
International Critical Tables of Numerical Data, Vol. III, p. 27–30, National Research Council of U.S.A. (McGraw-Hill, New York, 1928).
F.-M. Lee, L. E. Lahti and C. E. Stoops,J. Chem. Eng. Data 21, 36 (1976).
G. C. Benson and O. Kiyohara,J. Solution Chem. 9, 791 (1980).
J. L. Hales and J. H. Ellender,J. Chem. Thermodyn. 8, 1177 (1976).
TRCHP Tables, Selected Values of Properties of Hydrocarbons and Related Compounds, Vol. II (d), Thermodynamics Research Center Hydrocarbon Project, Texas A&M University (1973).
W. C. Schumb, C. N. Satterfield, and R. L. Wentworth, inHydrogen Peroxide (Reinhold, New York, 1955).
J. Timmermans, inPhysico-Chemical Constants of Pure Organic Compounds, Vol. I and II (Elsevier, New York, 1965).
TRC Tables, Selected Values of Properties of Chemical Compounds, Ref. 27, Vol. III (v). Thermodynamics Research Center Data Project, Texas A&M University (1966).
N. Nichols, R. Sköld, C. Spink, and I. Wadsö,J. Chem. Thermodyn. 8, 993 (1976).
M. Oguni and C. A. Angell,J. Chem. Phys. 73, 1948 (1980).
R. Lumry, C. Jolicoeur, E. Battistel, L.-L. Lemelin, and A. Anusien, To be published.
L. G. Hepler,Can. J. Chem. 47, 4613 (1969).
H. S. Frank, inWater: A Comprehensive Treatise, F. Franks, ed., Vol. I, (Plenum Press, New York, 1972).
R. Lumry, E. Battistel, and C. Jolicoeur,Trans. Faraday Soc., submitted.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Alary, J.F., Simard, M.A., Dumont, J. et al. Simultaneous flow measurement of specific heats and thermal expansion coefficients of liquids: Aqueoust-BuOH mixtures and neat alkanols and alkanediols at 25°C. J Solution Chem 11, 755–776 (1982). https://doi.org/10.1007/BF00650517
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00650517