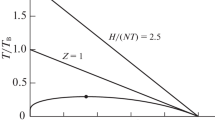
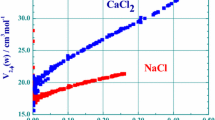
Abstract
Liquid systems which have strong non-idealities, as seen from their thermodynamic properties, often show evidence of these interactions in the solid-liquid phase diagrams. This suggests that some of the structures present in the solid state can persist in the solution state, on a time average, up to temperatures much higher than the melting point. Volumes and heat capacities of typical systems were either taken from the literature or measured to illustrate this correlation with the phase diagrams. With mixtures of aprotic solvents which show nearly-ideal simple eutectic phase diagrams, the properties of the solutions are also nearly ideal. Examples of systems investigated which show strong non-idealities are ionic surfactant solutions, alcohol-water mixtures, chloroform-triethylamine mixtures and lithium salts in aprotic solvents.
Similar content being viewed by others
References
G. Perron, L. Couture, F. Quirion, C. Camiré, and J. E. Desnoyers,J. Solution Chem. (in preperation).
G. Perron, L. Couture, D. Lambert, M. Hallé, and B. Marsan,Final Report, Phase II, June 1991, National Defense of Canada, Contract No 064 SSW2207-9-AF01.
L. G. Hepler, Z. S. Kooner, G. Roux-Desgranges, and J.-P. E. Grolier,J. Solution. Chem. 14, 579 (1985).
J.-P. E. Grolier, G. Roux-Desgranges, Z. S. Kooner, J. F. Smith, and L. G. Hepler,J. Solution Chem. 16, 745 (1985).
L. Barta, Z. S. Kooner, L. G. Hepler, G. Roux-Desgranges, and J.-P. E. Grolier,Can. J. Chem. 67, 1225 (1989).
F. Quirion, D. Lambert, and G. Perron,Can. J. Chem. (submitted).
P. Picker, E. Tremblay, and C. Jolicoeur,J. Solution. Chem. 3, 377 (1974).
P. Picker, P.-A. Leduc, P. Philip and, J. E. Desnoyers,J. Chem. Thermodyn. 3, 631 (1971).
J. Barthel, H.-J. Gores, G. Schmeer, and R. Wachter, inTopics In Current Chemistry Vol. 3, ed. F. L. Boschke, (Springer, Heidelberg 1983).
G. Caron, G. Perron, M. Lindheimer, and J. E. Desnoyers,J. Colloid & Interf. Sci. 106, 324 (1985).
J. W. Gershman,J. Phys. Chem. 61, 581 (1957).
P. Kékicheff, C. Grabielle-Madelmont, and M. J. Ollivon,J. Colloid & Interf. Sci. 131, 112 (1989).
F. Franks and J. E. Desnoyers, inWater Science Revs. I, F. Franks, ed., (Cambridge Univ. Press, New York, 1985), p. 171.
C. DeVisser, G. Perron, and J. E. Desnoyers,Can. J. Chem. 55, 856 (1977).
J. B. Ott, J. R. Goates, and B. A. Waite,J. Chem. Thermodyn. 11, 739 (1979).
A. H. Roux and J. E. Desnoyers,Proc. Ind. Acad. Sci. (Chem. Sci.) 98, 435 (1987).
G. W. Stapleton, M. Bellay, C. A. Wulff and L. G. Hepler,J. Chem. Eng. Data 11, 95 (1965).
Author information
Authors and Affiliations
Additional information
Paper written in the honor of Loren Hepler on the occasion of his retirement.
Rights and permissions
About this article
Cite this article
Perron, G., Couture, L. & Desnoyers, J.E. Correlation of the volumes and heat capacities of solutions with their solid-liquid phase diagrams. J Solution Chem 21, 433–443 (1992). https://doi.org/10.1007/BF00649697
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00649697