Abstract
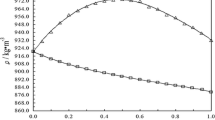
Densities and heat capacities of methanol + acetonitrile mixtures were determined over the whole composition range at 5 and 25°C. Apparent and partial molar volumes and heat capacities for both components of the mixture were calculated from these data. These functions for acetonitrile run monotonously over the whole composition range of the mixture at both temperatures, while those for methanol exhibit extrema (volumes-minimum and heat capacities-maximum) at high acetonitrile content. The reasons of the observed behavior of the system are discussed.
Similar content being viewed by others
References
A. Piekarska, H. Piekarski, and S. Taniewska-Osinska,J. Chem. Soc. Faraday Trans. I 82, 513 (1986).
S. Taniewska-Osinska and A. Piekarska,Thermochim. Acta (in press).
Y. Kondo, M. Ittoh, and S. Kusabayashi,J. Chem. Soc. Faraday Trans. I 78, 2793 (1982).
B. G. Cox and W. E. Waghorne,J. Chem. Soc. Faraday Trans. I 80, 1267 (1984).
C. Verhoef and E. Barendrecht,Anal. Chim. Acta 94, 395 (1977).
G. S. Kell,J. Chem. Eng. Data 12, 66 (1967).
P. Picker, C. Jolicoeur and J. E. Desnoyers,J. Chem. Educ. 45, 614 (1968).
H. C. Zegers, Thesis, Vrije Universiteit, Amsterdam (1988).
H. C. Zegers and G. Somsen,J. Chem. Thermodyn. 16, 225 (1984).
H. Piekarski and G. Somsen,J. Chem. Soc. Faraday Trans. I 84, 529 (1988).
H. C. Zegers, R. Boegschoten, W. Mels, and G. Somsen,Can. J. Chem. 64, 40 (1986).
G. T. Hefter, J.-P. E. Grolier, A. H. Roux, and G. Roux-Desranges,J. Solution Chem. 19, 207 (1990).
C. De Visser and G. Somsen,J. Solution Chem. 8, 593 (1978).
G. C. Benson and O. Kiyohara,J. Solution Chem. 9, 791 (1980).
G. C. Paraskevopoulos and R. W. Missen,Trans. Faraday Soc. I 58, 869 (1962).
N. Morel-Desrosiers and J.-P. Morel,J. Am. Chem. Soc. 103, 4743 (1981).
C. Moreau and G. DouheretThermochim. Acta 13, 385 (1975).
R. Zana, G. Perron, and J. E. Desnoyers,J. Solution Chem. 8, 729 (1979).
C. Luhrs and G. Schwitzgebel,Ber. Bunsenges. Phys. Chem. 83, 623 (1979).
Physico-Chemical Constants of Pure Organic Compounds, J. Timmermans, ed., (Elsevier, New York, 1950), p. 305.
W. E. Putnam, D. M. McEachern, and J. E. Kilpatrick,J. Chem. Phys. 42, 749 (1965).
D. D. Wagman, W. H. Evans, V. B. Parker, I. Halow, S. M. Bauley, and R. H. Schumm, Tech. Note 270-3, (Natl. Bureau Stds, Washington, D.C., 1968), p. 154; cited in Ref. 18.
C. De Visser, G. Perron, and E. Desnoyers,Can. J. Chem. 55, 856 (1977).
C. M. Slansky,J. Am. Chem. Soc. 62, 2430 (1940).
S. Taniewska-Osiriska and H. Piekarski,J. Solution Chem. 7, 891 (1978).
H. Piekarski,Can. J. Chem. 61, 2203 (1983).
Y. Pointud, J. Juillard, L. Avedikian, and J.-P. Morel,Thermochim. Acta 8, 423 (1974).
J. Lee, C. H. Kang, B.-S. Lee, and H. W. Lee,J. Chem. Soc. Faraday Trans. I 86, 1477 (1990).
H. PiekarskiCan J. Chem. 65, 2810 (1987).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Piekarski, H., somsen, G. Heat capacities and volumes of methanol+acetonitrile mixtures at 5 and 25°C. J Solution Chem 21, 557–567 (1992). https://doi.org/10.1007/BF00649564
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00649564