Abstract
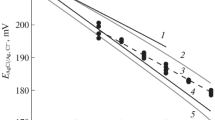
Reference values of the activity coefficient of hydrochloric acid at intervals of 5°C from 0 to 60°C are proposed. With their use, interlaboratory differences among silver—silver chloride electrodes can be eliminated.
Similar content being viewed by others
References
R. G. Bates and J. B. Macaskill,Pure Appl. Chem. 50, 1703 (1978).
R. G. Bates, E. A. Guggenheim, H. S. Harned, D. J. G. Ives, G. J. Janz, C. B. Monk, J. E. Prue, R. A. Robinson, R. H. Stokes, and W. F. K. Wynne-Jones,J. Chem. Phys. 25, 361 (1956);26, 222 (1957).
K. S. Pitzer and G. Mayorga,J. Phys. Chem. 77, 2300 (1973).
L. F. Silvester and K. S. Pitzer,J. Solution Chem. 7, 327 (1978).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bates, R.G., Robinson, R.A. Standardization of silver-silver chloride electrodes from 0 to 60°C. J Solution Chem 9, 455–456 (1980). https://doi.org/10.1007/BF00647735
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00647735