Abstract
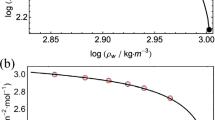
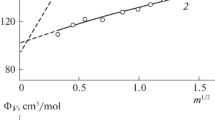
The ionization constant of orthophosphoric acid, determined by conductivity measurements, decreased from 7.11×10−3 at 25°C to 6.2×10−4 mol-kg−1 at 200°C. The pressure effect to 2000 bar was also measured and the ratio K2000/K1 is 2.7 at 25°C and 3.7 at 200°C. The standard partial molar volume change for the ionization at 1 bar,\(\Delta \overline V _{_1^o }\), changes from −16.1 at 25°C to −33.3 cm3-mol−1 at 200°C. The partial molar compressibility change for the ionization,\(\Delta \overline x ^\circ\), varies from −3.8×10−3 to −8.3×10−3 cm3-mol−1 bar−1 over the same temperature range.
Similar content being viewed by others
References
R. G. Bates,J. Res. Nat. Bur. Std. 47, 127 (1951).
L. F. Nims,J. Am. Chem. Soc. 56, 1110 (1934).
C. M. Mason and J. B. Culvern,J. Am. Chem. Soc. 71, 2387 (1949).
A. A. Noyes, Publication NO. 63, Carnegie Institution of Washington (1907).
J. M. Wright, W. T. Lindsay, Jr., and T. R. Druga, Westinghouse Atomic Power Division Report WAPD-TM-204, June 1961.
L. A. Palyuk and P. A. Kryukow,Izvestia Sibirsk. Otd. Akad. Nauk. SSR Ser. Khim. Nauk. 6, 88 (1978).
R. E. Mesmer and C. F. Baes Jr.,J. Solution Chem. 3, 307 (1974).
A. J. Ellis and D. W. Anderson,J. Chem. Soc. 1765 (1961).
A. Distèche and S. Dièsteche:J. Electrochem. Soc. 112, 350 (1965).
J. S. Smith, Dissertation, Yale University (1943).
A. Lo Surdo, K. Bernstrom, C. A. Jonsson, and F. J. Millero,J. Phys. Chem. 83, 1255 (1979).
J. W. Larson, K. G. Zeeb, and L. G. Hepler,Can. J. Chem. 60, 2141 (1982).
A. J. Read,J. Phys. E. Sci. Instru. 6, 694 (1973).
A. J. Read,J. Solution Chem. 4, 53 (1975).
A. J. Read,J. Solution Chem. 10, 437 (1981).
C. W. Childs, C. J. Downes, and R. F. Platford,Aust. J. Chem. 26, 863 (1973).
G. Olofsson and L. G. Hepler,J. Solution Chem. 4, 127 (1975).
A. S. Quist and W. L. Marshall,J. Phys. Chem. 69, 2984 (1965).
J. E. Smith and E. B. Dismukes,J. Phys. Chem. 68, 1603 (1964).
M. Selvaratnam and M. Spiro,Trans. Farad. Soc. 61, 360 (1965).
K. L. Elmore, J. D. Hatfield, R. L. Dunn, and A. D. Jones,J. Phys. Chem.,69, 3520 (1965).
C. W. Childs,J. Phys. Chem. 73, 2956 (1969).
C. M. Preston and W. A. Adams,J. Phys. Chem. 83, 814 (1979).
W. A. Adams, C. M. Preston, and H. A. M. Chew,J. Chem. Phys. 70, 2074 (1979).
A. J. Read, unpublished data.
A. B. Gancy and S. B. Brummer,J. Phys. Chem. 73, 2429 (1969).
L. Haar, J. S. Gallagher, and G. S. Kell,NBS/NRC Steam Tables (Hemisphere, New York, 1984).
F. H. Sweeton, R. E. Mesmer, and C. F. Baes Jr.,J. Solution Chem. 3, 191 (1974).
H. S. Harned and B. B. Owen,The Physical Chemistry of Electrolyte Solutions 3rd ed. (Reinhold, NY, 1958) p. 648.
A. J. Read,J. Solution Chem. 11, 629 (1982).
D. A. Lown, H. R. Thirsk, and W. F. K. Wynne-Jones,Trans. Faraday Soc. 66, 51 (1970).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Read, A.J. The first ionization constant from 25 to 200°C and 2000 bar for orthophosphoric acid. J Solution Chem 17, 213–224 (1988). https://doi.org/10.1007/BF00646175
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00646175