Abstract
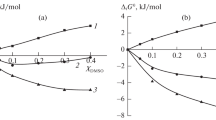
The kinetics of piperidine replacement by pyridine at the pentacyano(piperidine)-ferrate(II) complex ion was studied under pseudo-first order conditions in binary aqueous mixtures of methanol, t-butanol, p-dioxane, and glycerol, from a mole fraction of co-solvent from 0 to about 0.15. The observed variations can be explained considering the degree of hydrophobic interaction between released ligand and water molecules which changes according to the structure-forming or struucture-breaking effect of added co-solvent on water.
Similar content being viewed by others
References
M. J. Blandamer and J. Burgess,Coord. Chem. Rev. 31, 93 (1980).
G. C. Pedrosa, J. A. Salas, M. Katz, and N. E. Katz,J. Coord. Chem. (in press).
D. B. Soria, M. del V. Hidalgo, and N. E. Katz,J. Chem. Soc., Dalton Trans. 1555 (1982).
F. Franks, inHydrogen-bonded Solvent Systems, A. Covington and P. Jones, eds., (Taylor and Francis, London, 1968), p. 31.
G. C. Pedrosa, N. L. Hernández, N. E. Katz, and M. Katz,J. Chem. Soc., Dalton Trans. 2297 (1980).
J. A. Olabe and P. J. Aymonino,J. Inorg. Nucl. Chem. 38, 225 (1976).
D. P. Shoemaker and C. W. Garland,Experiments in Physical Chemistry, 2nd edn., (McGraw-Hill, New York, 1967).
N. E. Katz, P. J. Aymonino, M. A. Blesa, and J. A. Olabe,Inorg. Chem. 17, 556 (1978).
O. Kiyohara, G. Perron, and J. E. Desnoyers,Can. J. Chem. 53, 2591 (1975).
M. J. Blandamer, J. Burgess, and R. I. Haines,J. Chem. Soc., Dalton Trans. 385 (1976).
D. Oakenfull and D. E. Fenwick,J. Chem. Soc. Faraday Trans. I 636 (1979).
B. G. Cox,J. Chem. Soc., Perkin Trans. II 607 (1973).
F. Franks and D. J. G. Ives,Quart. Rev. 20, 1 (1966).
E. F. Caldin and H. P. Bennetto,J. Solution Chem. 2, 217 (1973).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Salas, J.A., Katz, M. & Katz, N.E. Hydrophobic interaction effects on the kinetics of ligand substitution in binary aqueous mixtures at pentacyano(piperidine)ferrate(II) ion. J Solution Chem 12, 115–122 (1983). https://doi.org/10.1007/BF00645352
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00645352