Abstract
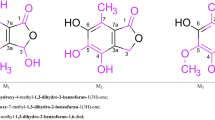
The paper is devoted to the synthesis of steroidal 5α-hydroxy-6-oxolactones from ethyl esters of 5,6-dienic acids. By epoxidating the latter it has been shown that, in addition to the usual formation of 5,6-oxides, opening of the 16α, 17-oxide ring initially formed takes place and this is accompanied by intramolecular cyclization to a 17,20-dihydroxy-β-lactone. The trans-opening of the 5α,6α-epoxide in the epoxy-δ-lactone and subsequent oxidation with N-bromosuccinimide has led to a new representative of steroidal δ-lactones — the 23,16-δ-lactone of the 3-acetate of 3β, 5α, 16β, 17α, 20ξ-pentahydroxy-6-oxo-24-norcholan-23-oic acid.
Similar content being viewed by others
Literature cited
A. V. Kamernitskii, V. A. Krivoruchko, R. P. Litvinovskaya, and I. G. Reshetova, Izv. Akad. Nauk SSSR, Ser. Khim., 2073 (1975).
K. Tori, T. Komeno, and T. Nakagawa, J. Org. Chem.,29, 1136 (1964).
E. Demole and H. Wuest, Helv. Chim. Acta,50, 1314 (1967).
A. V. Kamernitskii, V. A. Krivoruchko, and I. G. Peshetova, Izv. Akad. Nauk SSSR, Ser. Khim., 188 (1978).
P. Kocovsky and V. Cerny, Collect. Czech. Chem. Commun.,44, 1496 (1979).
M. Kocor and L. Tomaszewska, Bull. Acad. Polon. Sci.,23, 223 (1975).
Additional information
N. D. Zelinskii Institute of Organic Chemistry, Academy of Sciences of the USSR, Moscow. Translated from Khimiya Prirodnykh Soedinenii, No. 2, pp. 208–213, March–April, 1980.
Rights and permissions
About this article
Cite this article
Kamernitskii, A.V., Krivoruchko, V.A. & Reshetova, I.G. Transformed steroids 117. Synthesis of 5α-hydroxy-6-oxosteroids with a δ-lactone ring E. Chem Nat Compd 16, 162–166 (1980). https://doi.org/10.1007/BF00638777
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00638777