Abstract
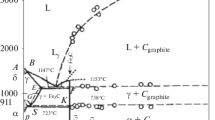
The kinetics of solid-state reactions of powdered reactants were investigated by X-ray and by differential thermogravimetry in a magnetic field. Measurements revealed mutual diffusion of the Fe3+ and In3+ ions in the Fe2O3-In2O3 system heat treated for 3 h at 700 to 1400° C. Diffusion of indium into the Fe2O3 lattice caused a shift of the Curie temperature of the antiferromagnetic iron oxide towards lower temperatures. Only Caln2O4 was found between CaCO3 and In2O3 up to 1400° C. Also, in the Fe2O3-CaCO3-In2O3in system, the reaction started with the mutual diffusion of iron and indium and the forming of CaFe2O4. End-products were the magnetic α-Ca4Fe14O25 and CaFe4O7, and the non-magnetic CaFe5O7, depending on the In3+ concentration. Indium stabilized the magnetic calcium-iron oxide structures, shifting their Curie temperatures towards lower values.
Similar content being viewed by others
References
A. Sztaniszlav, E. Sterk andL. Fetter,J. Magn. Magn. Mater. 41 (1984) 75.
P. B. Braun andW. Kwestroo,Philips Res. Rep. 15 (1960) 394.
B. Phillips andA. Muan,J. Amer. Ceram. Soc. 41 (1958) 445.
T. J. Malseva andV. J. Ljadova,Metalli 1 (1982) 6.
S. Holmquist,Nature 185 (1960) 604.
A. Sztaniszlav, M. Balla, M. Tardos, M. Farkas-Janke andJ. Sztatis, in Proceedings of the 7th International Conference on Microwave Ferrites, Smolenice, CSSR, September 1984, edited by O. Benda (Slovak Academy of Sciences, Bratislava, 1984) p. 116.
N. Ichinose,Jpn J. Appl. Phys. 5 (1966) 1140.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Sztaniszlav, A., Balla, M., Farkas-Jahnke, M. et al. Solid state reactions in the Fe2O3-CaCO3-In2O3 system. J Mater Sci 25, 2353–2358 (1990). https://doi.org/10.1007/BF00638027
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00638027