Summary
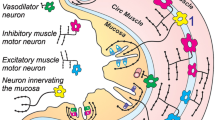
It is known that the majority of the mucosal nerve fibres in the guinea-pig small intestine arise from submucous ganglia. There are a number of neurochemically distinct populations of nerve cells in these ganglia, approximately half of them being cholinergic. In these studies we have stimulated isolated preparations of mucosa and submucosa with electrical field stimulation (EFS), 5-hydroxytryptamine (5-HT) and the nicotinic agonist 1,1-dimethyl-4-phenylpiperazinium (DMPP) and monitored changes in ion transport.
Segments of intestine were dissected free of external muscle and myenteric plexus and mounted in Ussing chambers. Short-circuit current (I sc) was measured as an indication of net ion transport across the tissue. EFS consisted of passing bipolar rectangular stimulus pulses through two platinum wires, one placed on each of the mucosal and submucosal sides of the tissue.
EFS, 5-HT and DMPP each caused a transient increase inI sc. Tetrodotoxin (TTX) abolished all of the EFS response and the majority of the response observed with 5-HT or DMPP, suggesting that the action of these stimuli on the mucosa is primarily nerve-mediated. The TTX-sensitive responses to 5-HT (>5×10−7 M) and DMPP consisted of two components, appearing with different latencies. The response to EFS also consisted of two components. Hyoscine abolished the first component of each of these responses and significantly reduced the amplitude of the second, by 40% (for EFS and 5-HT) and 84% (for DMPP). At lower 5-HT concentrations, only the later component was seen, and this was unaffected by hyoscine. These results suggest that the early component of each response is due to the release of acetylcholine from cholinergic nerves. The hyoscine-resistant responses to EFS and DMPP were reduced by a substance P antagonist (d-Arg1,d-Pro2,d-Trp7,9, Leu11), suggesting that these responses involve activation of substance P receptors in the mucosa.
The studies suggest that EFS and 5-HT (>5×10−7 M) stimulate both cholinergic and non-cholinergic nerves effectively, that 5-HT (10−8–5×10−7 M) preferentially stimulates non-cholinergic nerves and that DMPP preferentially stimulates cholinergic nerves.
Similar content being viewed by others
References
Cassuto J, Jodal M, Tuttle R, Lundgren O (1982) 5-Hydroxytryptamine and cholera secretion. Physiological and pharmacological studies in cats and rats. Scand J Gastroenterol 17:695–703
Cooke HJ (1984) Influence of enteric cholinergie neurons on mucosal transport in guinea-pig ileum. Am J Physiol 246: G263-G267
Cooke HJ, Carey HV (1984) The effects of cisapride on serotonin-evoked mucosal responses in guinea-pig ileum. Eur J Pharmacol 98:147–148
Cooke HJ, Shonnard K, Wood JD (1983) Effects of neuronal stimulation on mucosal transport in guinea pig ileum. Am J Physiol 245:G290-G296
Costa M, Furness JB, Cuello AC, Verhofstad AAJ, Steinbusch HWJ, Elde RP (1982) Neurons with 5-hydroxytryptamine-like immunoreactivity in the enteric nervous system: Their visualization and reactions to drug treatment. Neuroscience 7:351–363
Costa M, Furness JB, Pullin CO, Bornstein J (1985) Substance P enteric neurons mediate non-cholinergic transmission to the circular muscle of the guinea-pig intestine. Naunyn-Schmiedeberg's Arch Pharmacol 328:446–453
Donowitz M, Charney AN, Heffernan JM (1977) Effect of serotonin treatment on intestinal transport in the rabbit. Am J Physiol 232:E85-E94
Donowitz M, Tai YH, Asarkof N (1980) Effect of serotonin treatment on active electrolyte transport in rabbit ileum, gallbladder and colon. Am J Physiol 239:G463-G472
Furness JB, Costa M, Keast JR (1984) Choline acetyltransferase and peptide immunoreactivity of submucous neurons in the small intestine of the guinea-pig. Cell Tiss Res 237:328–336
Furness JB, Costa M, Gibbins IL, Llewellyn-Smith IJ, Oliver JR (1985) Neurochemically similar myenteric and submucous neurons directly traced to the mucosa of the small intestine. Cell Tiss Res 241:155–163
Gaginella TS, O'Dorisio TM, Hubel KA (1981) Release of vasoactive intestinal polypeptide by clectrical field stimulation of rabbit ileum. Regul Pept 2:165–174
Gershon MD (1967) Effects of tetrodotoxin on innervated smooth muscle preparations. Br J Pharmac Chemother 29:259–279
Hardcastle J, Hardcastle PT, Redfern JS (1981) Action of 5-hydroxytryptamine on intestinal ion transport in the rat. J Physiol 320:41–55
Hirst GDS, McKirdy HC (1975) Synaptic potentials recorded from neurones of the submucous plexus of guinea-pig small intestine. J Physiol 249:369–386
Hirst GDS, Silinsky EM (1975) Some effects of 5-hydroxytryptamine, dopamine and noradrenaline on neurons in the submucous plexus of guinea-pig small intestine. J Physiol 251:817–832
Hubel KA (1978) The effects of electrical field stimulation and tetrodotoxin on ion transport by the isolated rabbit ileum. J Clin Invest 62:1039–1047
Hubel KA, Shirazi S (1982) Human ileal ion transport in vitro: changes with electrical field stimulation and tetrodotoxin. Gastroenterology 83:63–68
Keast JR, Furness JB, Costa M (1984) The origins of peptide and norepinephrine nerves in the mucosa of the guinea-pig small intestine. Gastroenterology 86:637–644
Keast JR, Furness JB, Costa M (1985) Different substance P receptors are found on mucosal epithelial cells and submucous neurons of the guinea-pig small intestine. Naunyn-Schmiedeberg's Arch Pharmacol 329:382–387
Kilbinger H, Stauss P, Holzer P (1984) Antagonist discrimination between neuronal and muscular receptors for substance P in the guinea-pig ileum. IUPHAR 9th Int Congress Pharmacol, London, p 184, Macmillan Press, London
Kisloff B, Moore EW (1976) Effect of serotonin on water and electrolyte transport in the in vivo rabbit small intestine. Gastroenterology 71:1033–1038
Kuriyama H, Osa T, Toida N (1966) Effects of tetrodotoxin on smooth muscle cells of the guinea-pig taenia coli. Br J Pharmacol 27:366–376
Nemeth PR, Ewart WR, Wood JD (1983) Effects of the putative substance P antagonists (d-Pro2,d-Phe7,d-Trp9, andd-Pro2,d-Trp7,d-Trp9) on electrical activity of myenteric neurons. J Autonom Nerv Sys, Suppl. 8:165–169
Rothe CF, Quay JF, Armstrong WM (1969) Measurement of epithelial electrical characteristics with an automatic voltage clamp device with compensation for solution resistance. IEEE Trans Biomed Eng 4:160–164
Schultz SG, Zalusky R (1964) Ion transport in isolated rabbit ileum. I. Short-circuit current and Na fluxes. J Gen Physiol 47:567–584
Sheerin HE (1979) Serotonin action on short-circuit current and ion transport across isolated rabbit ileal mucosa. Life Sci 24:1609–1616
Tapper EJ, Lewand DL (1981) Actions of a nicotinic agonist, DMPP, on intestinal ion transport in vitro. Life Sci 28:155–162
Tapper EJ (1983) Local modulation of intestinal ion transport by enteric nerves. Am J Physiol 244:G457-G468
Ussing H, Zerahn K (1951) Active transport of sodium as the source of electric current in the short-circuited isolated frog skin. Acta Physiol Scand 23:110–127
Zimmerman TW, Binder HJ (1983) Effect of tetrodotoxin on cholinergic agonist-mediated colonic electrolyte transport. Am J Physiol 244:G386-G391
Zimmerman TW, Binder HJ (1984) Serotonin-induced alteration of colonic electrolyte transport in the rat. Gastroenterology 86:310–317
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Keast, J.R., Furness, J.B. & Costa, M. Investigations of nerve populations influencing ion transport that can be stimulated electrically, by serotonin and by a nicotinic agonist. Naunyn-Schmiedeberg's Arch. Pharmacol. 331, 260–266 (1985). https://doi.org/10.1007/BF00634247
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00634247