Abstract
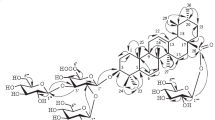
From the epigeal part ofThalictrum minus L. (Ranuncluaceae) we have isolated a minor cycloartane trisdesmoside — thalicoside C — 3β,16β,22(S),29-tetrahydroxy-9,19-cyclo-20(S)-lanost-24-ene 3-O-β-galactopyranoside 22,29-di-O-β-D-glucopyranoside. Its structure was established from the results of chemical transformations and of FAB-mass and NMR spectroscopies (1H1,13C,1H-1H1, and13C-1H).
Similar content being viewed by others
Literature cited
A. S. Gromova, V. I. Lutskii, A. A. Semenov, V. A. Denisenko, and V. V. Isakov, Khim. Prir. Soedin., 213 (1984).
A. S. Gromova, V. I. Lutskii, A. A. Semenov, R. B. Valeev, G. A. Kalabin, and Yu. N. El'kin, Khim. Prir. Soedin., 670 (1985).
A. S. Gromova, V. I. Lutskii, A. L. Vereshchagin, and A. A. Semenov, Khim. Prir. Soedin., 107 (1987).
S. Seo, G. Tomita, K. Tori, and G. Goshimura, J. Am. Chem. Soc.,100, 3331 (1978).
A. Bax and D. G. Davis, J. Magn. Reson.,65, 355 (1985).
M. Levitt, R. Freeman, and T. Frenkell, J. Magn. Reson.,47, 328 (1982).
Additional information
Irkutsk Institute of Organic Chemistry, Siberian Branch, Russian Academy of Sciences. Translated from Khimiya Prirodnykh Soedeninii, No. 1, pp. 103–109, January–February, 1993.
Rights and permissions
About this article
Cite this article
Gromova, A.S., Lutskii, V.I., Zinchenko, S.V. et al. Triterpene saponins from Thalictrum minus. VI. Structure of thalicoside C. Chem Nat Compd 29, 87–91 (1993). https://doi.org/10.1007/BF00631029
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00631029