Abstract
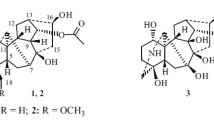
The structure of the new diterpene alkaloid acofine, isolated from tubers ofAconitum karakolicum, has been shown by the x-ray structural method (diffractometer, CuKα, 1250 reflections, direct method, R=0.071). The IR, mass, and PMR spectrum of acofine and its monoacetate are considered. Acofine is the first chlorine-containing diterpene alkaloid, and also the first alkaloid of the napelline type with a 15,16β-acetonide group.
Similar content being viewed by others
Literature cited
M. N. Sultankhodzhaev, M. S. Yunusov, and S. Yu. Yunusov, Khim. Prir. Soedin., 199 (1973).
A. A. Nishanov, B. Tashkhodzhaev, I. M. Yusupova, and M. N. Sultankhodzhaev, Khim. Pr. Soedin., 534 (1992).
N. Batbayar, D. Batsurén, B. Tashkhodzhaev, M. I. Yusupova, and M. N. Sultankhodzhaev, Khim. Pr. Soedin., No. 1, 60 (1993).
F. H. Allen, O. Kennard, D. G. Watson, L. Bramer, A. G. Orpen, and R. Taylor, J. Chem. Soc., Perkin Trans. II, No. 12, S1–S19 (1987).
B. Tashkhodzhaev, Khim. Pr. Soedin., 231 (1982).
M. N. Benn, F. Oranga, and J. F. Richardson, Heterocycles,26, 2331 (1987).
G. M. Sheldrick, SHELXS-86 Program for Crystal Structure Determination, Gottingen, GFR.
G. M. Sheldrick, SHELX-76 Program for Crystal Structure Determination, Cambridge University Press, Cambridge, UK (1976).
Additional information
Institute of the Chemistry of Plant Substances, Uzbekistan Academy of Sciences, Tashkent. Translated from Khimiya Prirodnykh Soedinenii, No. 2, pp. 267–272, March–April, 1993.
Rights and permissions
About this article
Cite this article
Tashkhodzhaev, B., Sultankhodzhaev, M.N. & Yusupova, I.M. Structure of the new unusual diterpene alkaloid acofine from Aconitum karakolicum. Chem Nat Compd 29, 222–225 (1993). https://doi.org/10.1007/BF00630121
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00630121