Abstract
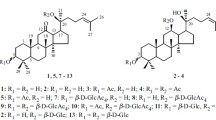
The products of the interaction of α-acetobromoglucose with 2,4,5-trihydroxyphenyl benzyl ketone and 2,4-dihydroxyphenyl 4′-hydroxybenzyl ketone and also with their heterocyclic analogues, which are present completely or partially in the enolic form, have been investigated. It has been shown that in this reaction a tetra-O-acetylglucosyl residue is added at the 4-OH hydroxy group of the phenyl residue of the ketone. The compounds obtained have been converted into isoflavone 7-O-glucosides by the action of the vilsmeier reagent or acetoformic anhydride.
Similar content being viewed by others
Literature cited
V. G. Pivovarenko, V. P. Khilya, V. N. Kovalev, and S. A. Vasil'ev, Khim. Prir. Soedin., No. 4, 511 (1988).
V. G. Pivovarenko, V. P. Khilya, V. N. Kovalev, and S. A. Vasil'ev, Khim. Prir. Soedin., 519 (1988).
A. L. Kazakov, V. P. Khilya, V. V. Mezheritskii, and Yu. Litken, Natural and Modified Isoflavonoids [in Russian], Izd. RGU, Rostov-on-Don (1985).
V. N. Kovalev, N. F. Komissarenko, V. P. Khilya, L. D. Khaleeva, and I. N. Timasheva, in: Basic Directions of Pharmacy: Abstracts of Lectres at the IInd Congress of Pharmacists of the Latvian SSR [in Russian], Riga (1984), p. 303.
V. Szabo, J. Borbely, and J. Borda, in: Proceedings of an International Bioflavonoid Symposium, Munich, FRG (1981), p. 19.
V. P. Khilya, T. M. Tkachuk, I. P. Kupchevskaya, and G. M. Golubushkina, Dokl. Akad. Nauk UkrSSR, Ser. B, No. 5, 61 (1980).
I. P. Kupchevskaya and V. Khilya, Dokl. Akad. Nauk UkrSSR, Ser. B, No. 2, 119 (1979).
V. G. Pivovarenko and V. P. Khilya, Khim. Geterotsikl. Soedin., 625 (1991).
L. Farkas and A. Wolfner, Acta Chim. Acad. Sci. Hung.,67, No. 1, 109 (1971).
L. Farkas and J. Varady, Chem. Ber.,92, No. 4, 819 (1959).
Additional information
Taras Shevchenko Kiev University. Translated from Khimiya Prirodnykh Soedinenii, No. 2, pp. 220–227, March–April, 1993.
Rights and permissions
About this article
Cite this article
Pivovarenko, V.G., Khilya, V.P. Synthesis of 7-O-β-D-glucopyranosides of isoflavones and their heterocyclic analogues. Chem Nat Compd 29, 181–186 (1993). https://doi.org/10.1007/BF00630111
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00630111