Summary
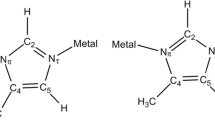
The enthalpies of the first step of complex formation of 1-methylimidazole with CoII, NiII, CuII and ZnII have been determined by direct calorimetry. By means of the equilibrium constants, the Gibbs functions and entropies were also obtained. The measurements were carried out in aqueous medium, at 25 °C and an ionic strength I=1 mol dm−3 (NaNO3). The present results and the previous thermodynamic data for associations of pyrazole and imidazole are used to infer the quality of the metal-ligand bonds and particularly their π-character.
Similar content being viewed by others
References
R. Aruga,J. Inorg. Nucl. Chem., 43, 2459 (1981).
J. E. Bauman Jr. and J. C. Wang,Inorg. Chem., 3, 368 (1964).
J. J. Christensen, D. J. Eatough and R. M. Izatt,Handbook of Metal Ligand Heats, Marcel Dekker, New York, 1975.
B. Lenarcik and B. Barszcz,Rocz. Chem., 51, 1849 (1977).
Y. Nozaki, F. R. Gurd, R. F. Chen and J. T. Edsall,J. Am. Chem. Soc., 79, 2123 (1957).
R. Aruga,Aust. J. Chem., 34, 501 (1981).
M. A. Marini and R. L. Berger,Anal. Biochem., 43, 188 (1971).
L. D. Hansen, E. J. Baca and P. Scheiner,J. Heterocyclic Chem., 7, 991 (1970).
G. Anderegg,Helv. Chim. Acta, 43, 414 (1960).
R. Aruga,J. Inorg. Nucl. Chem., 41, 845 (1979).
M. S. Sun and D. G. Brewer,Can. J. Chem., 45, 2729 (1967).
L. G. Sillén,Acta Chem. Scand., 16, 159 (1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Aruga, R. Thermodynamics of complex formation of 1-methylimidazole with cobalt(II), nickel(II), copper(II) and zinc(II) cations in aqueous solution. Transition Met Chem 8, 56–58 (1983). https://doi.org/10.1007/BF00618801
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00618801