Abstract
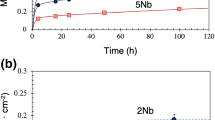
The aligned eutectic alloy Ni3Al -Ni3Nb at 1155°C exhibits an initial very rapid rate of oxidation which rate eventually decreases to a very significantly lower level. This decrease in the rate of oxidation has been shown to be associated with the formation of an aluminum oxide layer. The work reported here was conducted to determine both how such a layer forms in oxidation at 1155°C as a function of time and which elements are the active diffusing species in the oxidation process.
Similar content being viewed by others
References
J. G. Smeggil and M. D. McConnell,Oxid. Metals 8, 309 (1974).
M. P. Arbuzov and V. G. Chuprina,Izv. Akad. Nauk SSSR, Neorg. Mat. 1(7), 1121 (1965), Engl. transl.
M. P. Arbuzov and V. G. Chuprina,Izv. VUZ. Fizika, No. 12, 75 (1969), Engl. transl.
U. R. Evans,Rev. Pure Appl. Chem. 5(1), 1 (1955).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Smeggil, J.G. An examination of the mechanisms involved in the oxidation of the aligned eutectic alloy Ni3Al-Ni3Nb. Oxid Met 9, 31–44 (1975). https://doi.org/10.1007/BF00613493
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00613493