Abstract
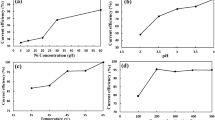
The effect ofgermanium on the electrowinning of zinc from industrial acid sulphate electrolyte was studied using X-ray diffraction, scanning electron microscopy and cyclic voltammetry techniques. Germanium concentrations > 0.1 mgl−1 results in severe re-solution of the zinc deposit and hence decreased the zinc deposition current efficiency. Extreme fluctuations in the current efficiency occurred as a function of electrolysis time. Cyclic voltammograms obtained for Ge-containing electrolytes were characterized by a shoulder in the reverse scan prior to the cross-over potential. Vigorous hydrogen gassing occurred at the shoulder. These results are interpreted in terms of the formation of local Zn-Ge galvanic cells. Germanium concentrations to 0.2 mgl−1 had no effect on the morphology of the 1-h zinc deposits but the preferred orientation changed from [1 1 4] [1 1 2] for Ge-free electrolyte to [1 1 2] [1 1 0] for electrolytes containing Ge.
Similar content being viewed by others
References
D. R. Fosnacht and T. J. O'Keefe,J. Appl. Electrochem. 10 (1980) 495.
M. Maja, N. Penazzi, R. Fratesi and G. Roventi,J. Electrochem. Soc. 129 (1982) 2695.
D. R. Fosnacht, PhD thesis, University of Missouri-Rolla (1979).
D. J. MacKinnon and J. M. Brannen,J. Appl. Electrochem. 7 (1977) 451.
D. J. MacKinnon, J. M. Brannen and R. C. Kerby,ibid. 9 (1979) 55.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Mackinnon, D.J., Fenn, P.L. The effect of germanium on zinc electrowinning from industrial acid sulphate electrolyte. J Appl Electrochem 14, 467–474 (1984). https://doi.org/10.1007/BF00610811
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00610811