Abstract
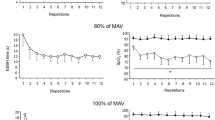
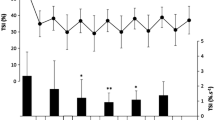
Anaerobic energy capacity was evaluated by maximal oxygen deficit (MOD) as well as by blood gas and muscle biopsy variables during short exhausting running in six recreational (RR) and eight competitive sprint and middle distance runners (SMDR). On 3 days runs to exhaustion were executed. Two runs were performed at a treadmill gradient of 15% at speeds which resulted in exhaustion after approximately 1 (R15%, 1min) and 2–3 min (R15%, 2–3min), respectively. On the 3rd day, the subjects ran with the treadmill at a gradient of 1% at a speed which caused exhaustion after 2–3 min (R1%, 2–3min). The runner performance was assessed from 400 m [RR, median 64.8 (range 62.2–69.6) s; SMDR, median 49.4 (range 48.5–52.0) s] and 800 m [RR, median 158.8 (range 153.3–170.2) s; SMDR, median 115.2 (range 113.3–123.3) s] track times. Muscle biopsies from gastrocnemius muscle were obtained before and immediately after R15%, 2–3min, from which muscle lactate and creatine phosphate (CP) concentrations, fibre type distribution, capillaries per fibre, total lactate dehydrogenase (LDH) activity and the LDH isoenzyme pattern were determined. The MOD increased with the treadmill gradient and duration. During both treadmill and track runs, SMDR performance was superior to that of RR, but no significant differences were observed with respect to MOD, muscle fibre type distribution, total LDH activity, its iso-enzyme pattern, changes in muscle lactate or CP concentrations. However, after treadmill runs, peak venous lactate concentration and partial pressures of carbon dioxide were higher, and pH lower in SMDR. Also the number of capillaries per muscle fibre and the maximal oxygen uptake were larger in SMDR. These findings would suggest that the superior performance of SMDR depended more on their aerobic than on their anaerobic capacity.
Similar content being viewed by others
References
Andersen P (1975) Capillary density in skeletal muscle of man. Acta Physiol Scand 95:203–205
Bangsbo J, Gollnick PD, Graham TE, Juel C, Kiens B, Mizuno M, Saltin B (1990) Anaerobic energy production and O2 deficit-debt relationship during exhaustive exercise in humans. J Physiol (Lond) 422:539–559
Brooke MH, Kaiser KK (1970) Three “myosin ATPase” systems: the nature of their pH lability and sulfhydryl dependence. J Histochem Cytochem 18:670–672
Cheetham ME, Boobis LH, Brooks S, Williams C (1986) Human muscle metabolism during sprint running. J Appl Physiol 61:54–60
Costill DL, Jansson E, Gollnick PD, Saltin B (1974) Glycogen utilization in leg muscles of men during level and uphill running. Acta Physiol Scand 91:475–481
Costill DL, Daniels J, Evans W, Fink W, Krahenbuhl G, Saltin B (1976) Skeletal muscle enzymes and fiber composition in male and female track athletes. J Appl Physiol 40:149–154
Durnin JV, Womersley J (1974) Body fat assessed from total body density and its estimation from skinfold thickness: measurements on 481 men and women aged from 16 to 72 years. Br J Nutr 32:77–97
Henson LC, Poole DC, Whipp BJ (1989) Fitness as a determinant of oxygen uptake response to constant-load exercise. Eur J Appl Physiol 59:21–28
Juel C, Bangsbo J, Graham T, Saltin B (1990) Lactate and potassium fluxes from skeletal muscle during intense dynamic knee-extensor exercise in man. Acta Physiol Scand 104:1–9
Karlsson J (1971) Lactate and phosphagen concentrations in working muscle of man. Acta Physiol Scand [Suppl] 358:1–72
Karlsson J, Hultén B, Sjödin B (1974) Substrate activation and product inhibition of LDH activity in human skeletal muscle. Acta Physiol Scand 92:21–26
Karlsson J, Sjödin B, Thorstensson A, Hultén B, Frith K (1975) LDH isozymes in skeletal muscle of endurance and strength trained athletes. Acta Physiol Scand 93:150–156
Keiding R, Hörder M, Gerhardt W (1974) Recommended methods for the determination of four enzymes in blood. Scan J Clin Lab Invest 33:291–306
Lowry OH, Passonneau JV (1972) A flexible system on enzymatic analysis. Academic Press, New York, pp 93–108
Medbø JI, Burgers S (1990) Effect of training on the anaerobic capacity. Med Sci Sports Exerc 22:501–507
Medbø JI, Mohn AC, Tabata I, Bahr R, Vaage O, Seijersted OM (1988) Anaerobic capacity determined by maximal accumulated O2 deficit. J Appl Physiol 64:50–60
Nevill ME, Boobis LH, Brooks S, Williams C (1989) Effect of training on muscle metabolism during treadmill sprinting. J Appl Physiol 67:2376–2382
Olesen HL (1992) Accumulated oxygen deficit increases with inclination of uphill running. J Appl Physiol 73:1130–1134
Padykula HA, Herman E (1955) The specificity of the histochemical method of adenosine triphosphatase. J Histochem Cytochem 3:170–195
Pilegaard H, Juel C, Wibrand F (1993) Lactate transport studied in sacrolemmal giant vesicles from rats: effect of training. Am J Physiol 264:E156-E160
Poole DC, Schaffartzik W, Knight DR, Derion T, Kennedy B, Guy HJ, Predileletto R, Wagner PD (1991) Contribution of exercising legs to the slow component of oxygen uptake kinetics in humans. J Appl Physiol 71:1245–1253
Raabo E (1963) Colorimetric method for demonstrating lactic dehydrogenase isoenzymes separated by paper electrophoresis. Scan Clin Lab Invest 15:405–414
Rasmussen J, Hanel B, Diamant B, Secher NH (1991) Muscle mass effect on arterial desaturation after maximal exercise. Med Sci Sports Exerc 23:1349–1352
Roston WL, Whipp BJ, Davis JA, Cunningham DA, Effros RM, Wasserman K (1987) Oxygen uptake kinetics and lactate concentration during exercise in humans. Am Rev Respir Dis 135:1080–1084
Saltin B, Gollnick PD (1983) Skeletal muscle adaptability significance for metabolism and performance. In: Peachey LD, Adrian PH, Geiger SR (eds) Handbook of physiology; skeletal muscle. American Physiology Society, Bethesda, pp 555–631
Scott CB, Roby FB, Lohman TG, Bunt JC (1991) The maximally accumulated oxygen deficit as an indicator of anaerobic capacity. Med Sci Exerc 23:618–624
Secher NH, Ruberg-Larsen N, Binkhorst RA, Bonde-Petersen F (1974) Maximal oxygen uptake during arm cranking and combined arm plus leg exercise. J Appl Physiol 36:515–518
Secher NH, Clausen JP, Klausen K, Noer I, Trap-Jensen J (1977) Central and regional circulatory effects of adding arm exercise to leg exercise. Acta Physiol Scand 100:288–297
Sjödin B, Thorstensson A, Frith K, Karlsson J (1976) Effect of physical training on LDH activity and LDH isozyme pattern in human skeletal muscle. Acta Physiol Scand 97:150–157
Tesch P (1979) Muscle fatigue in man. Acta Physiol Scand [Suppl] 480:1–40
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Olesen, H.L., Raabo, E., Bangsbo, J. et al. Maximal oxygen deficit of sprint and middle distance runners. Europ. J. Appl. Physiol. 69, 140–146 (1994). https://doi.org/10.1007/BF00609406
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00609406