Abstract
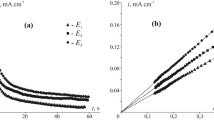
The anodic behaviour of zinc in 8 M NaOH/1 M Na2Zn(OH)4, containing 0.1, 1 and 10 v/o sodium silicate solution, has been studied under quiescent conditions at ambient temperature using cyclic linear sweep voltammetry in conjunction with darkfield illumination optical photo-microscopy.
Results, together with measurements of the double layer capacitance of the zinc/alkali interphase as a function of potential, suggest the presence of a surface film of silicate which induces early passivation in the anodic potential excursion.
Similar content being viewed by others
References
V.N. Flerov,Zh. Prikl. Khim. 30 (1957) 1326.
V.N. Flerov,ibid 31 (1957) 49.
V.V. Bakaev, V.N. Shirokov and V.N. Flerov,Electrokhimiya 7 (1971) 376.
A. Marshall, N.A. Hampson and J.S. Drury,J. Electroanal. Chem. 50 (1974) 292.
R.W. Powers,Electrochem. Technol. 5 (1967) 429.
R.W. Powers and M.W. Breiter,J. Electrochem. Soc. 116 (1969) 719.
R.W. Powers,ibid 1652.
J.J. McMullen and N. Hackerman,ibid 106 (1959) 341.
K. Huber,Helv. Chim. Acta. 26 (1943) 1037.
K. Huber,J. Electrochem. Soc. 100 (1953) 376.
D.A. Vermilyea, Anodic Films. Chapt. IV, Vol III,Adv. Electrochem. Eds. P. Delahay and C.W. Tobias, Interscience (1963).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Lewis, R.W., Turner, J. The effect of silicate ion on the anodic behaviour of zinc in concentrated alkali. J Appl Electrochem 5, 343–349 (1975). https://doi.org/10.1007/BF00608798
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00608798