Abstract
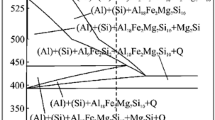
The morphology and composition of the oxide film formed under various oxidation conditions on a 1.2% aluminum-magnesium alloy have been studied using electron microscopy and diffraction. Below 350° C the oxide film formed is highly protective and amorphous, while above this temperature the oxide becomes nonprotective. Initially, at temperatures above 350°C, the oxide film consists of only alumina, then with increasing time it contains first MgO, then MgO together with MgAl2O4, and finally it is entirely converted to MgAl2O4.
Similar content being viewed by others
References
K. Haufle,Progr. Metal Phys. 4, 71–104 (1953).
C. Wagner,J. Electrochem. Soc. 99, 369–380 (1952).
W. E. Boggs, R. H. Kachik, and G. E. Pellissier,J. Electrochem. Soc. 110, 4–11 (1963).
M. Hansen,Constitution of Binary Alloys (McGraw-Hill, New York, 1958), p. 106.
W. W. Smeltzer,J. Electrochem. Soc. 105, 67–71 (1958).
D. M. Poole and H. J. Axon,J. Inst. Metals 80, 599–604 (1951–52).
K. Thomas and M. W. Roberts,J. Appl. Phys. 32, 70–75 (1961).
A. F. Beck, M. A. Heine, E. J. Caule, and M. J. Pryor,Corrosion Sci. 7, 1–22 (1967).
J.A.N.A.F. Thermochemical Tables (1965–66).
D. R. Spalding, J. W. Edington, R. E. Villagrana,Phil. Mag. 20, 1203–1212 (1969).
S. R. J. Saunders and M. J. Pryor,J. Electrochem. Soc. 115, 1037–1039 (1968).
G. L. Hunt and I. M. Ritchie,Oxidation of Metals 2, 361–371 (1970).
J. E. Castle, S. J. Gregg, and W. B. Jepson,J. Electrochem. Soc. 109, 1018–1023 (1962).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Ritchie, I.M., Sanders, J.V. & Weickhardt, P.L. Oxidation of a dilute aluminum magnesium alloy. Oxid Met 3, 91–101 (1971). https://doi.org/10.1007/BF00604741
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00604741