Abstract
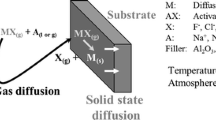
Six alloys, 310 stainless steel, Hastelloy X, Inconel 671, Incoloy 800, Haynes 188, and FeCrAlY (GE1541 and MA956), were corroded in two chars at 1600° and 1800°F. The chars, FMC and Husky, contained 2.7% and 0.9% sulfur, respectively. Various parameters were investigated, including char size, cover gas, char quantity, char replenishment period, gas composition, and the use of coatings. The corrosion process was strictly sulfidation when the char was replenished every 24 hr or less. The kinetics of reaction were nearly linear with time. The reaction resulted in thick external sulfide scales with extensive internal sulfidation in the substrate. The kinetics and reactionproduct morphologies suggested that diffusion through the sulfide scale played a minor role, and that an interfacial reaction was the rate-controlling step, A mathematical model was developed which supported this hypothesis.
The reaction rates showed a relatively minor role on alloy composition, depending upon whether the alloys were tested singularly or in combination with others. Inconel 671, the best alloy in CGA environments, consistently corroded the most rapidly of the chromia-former types regardless of char sulfur content or of the temperature. Type 310 stainless was marginally better than Inconel 671. Incoloy 800 was intermediate, whereas, Haynes 188 and Hastelloy X exhibited the best corrosion resistance. The FeCrAlY alloys reacted very rapidly in the absence of preoxidation treatments. All alloys corroded in char at least 1000 times more rapidly than in the CGA (MPCITTRI) environment. None of the alloys will be acceptable for use in contact with char unless coatings are applied.
Similar content being viewed by others
References
L. S. Darken and R. W. Gurry,Physical Chemistry of Metals (McGraw-Hill, New York, 1953).
H. K. Kobayoshi and C. Wagner,J. Chem. Phys. 26 1609 (1957).
E. T. Turkdogan, W. N. McKewan, and L. Zwell,J. Phys. Chem. 69 327 (1965).
A. O. Schaefer, “A Program to Discover Materials Suitable for Service under Hostile Conditions Obtaining in Equipment for the Gasification of Coal and other Solid Fuels,” FE-1784-33, Quarterly Progress Report, Metal Properties Council, December 1977.
E. B. Backensto, R. D. Drew, and C. C. Stapleford,Corrosion 12 22 (1956).
R. B. Setterlund and G. R. Prescott,Corrosion 17 103 (1961).
E. J. Vineberg, Ph.D. thesis, School of Engineering and Applied Science, UCLA (1979).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Douglass, D.L., Bhide, V.S. & Vineberg, E. The corrosion of some superalloys in contact with coal chars in coal gasifier atmospheres. Oxid Met 16, 29–79 (1981). https://doi.org/10.1007/BF00603744
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00603744