Abstract
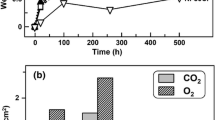
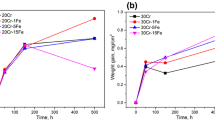
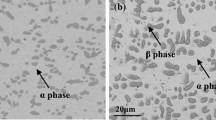
The oxidation of TD NiCr (Ni-20 wt. % Cr-2ThO2) has been investigated between 900 and 1200°C, and the oxidation behavior is compared with Ni-30 wt. % Cr and Co-35 wt. % Cr alloys. All alloys develop Cr2O3 scales but the weight changes obtained for the NiCr and CoCr alloys show an increase with time whereas above 1000° C the TD NiC shows a progressive loss in weight from the evaporation of CrO3 from the scale, and the reaction products appear to be formed mainly at the alloy-scale interface. However, no mechanism for its formation has been established.
Similar content being viewed by others
References
R. Irmann,Aluminium 27, (1951).
F. S. Pettit and E. J. Felton,J. Electrochem. Sac. 111, 135 (1964).
D. A. Jones and R. E. Westerman,Corrosion 21, 295 (1965).
W. C. Hagel,Trans. ASM 56, 583 (1963).
B. A. Wilcox, A. H. Clauer, and W. S. McCain,Trans. AIME 239, 1791 (1967).
G. S. Giggins and F. S. Pettit,Trans. AIME 245, 2495 (1969).
P. K. Kofstad and A. Z. Hed,J. Electrochem. Soc. 116, 224 (1969).
P. K. Kofstad and A. Z. Hed,Werkstoffe Korrosion (to be published).
S. A. Jansen and E. A. Gulbransen, 4th International Congress on Metallic Corrosion, September, 1969.
G. C. Graham and H. H. Davies,J. Am. Ceram. Soc. (to be published).
G. S. Giggins and F. S. Pettit,Trans. AIME 245, 2509 (1969).
M. J. Fleetwood,J. Inst. Metals 94, 218 (1966).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wallwork, G.R., Hed, A.Z. The oxidation of Ni-20 wt. % Cr-2ThO2 . Oxid Met 3, 229–241 (1971). https://doi.org/10.1007/BF00603523
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00603523