Abstract
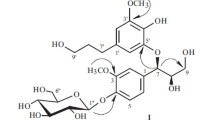
On the basis of chemical transformations and with the aid of physicochemical results, the structure of glycoside I isolated from the roots of the plantMedicago sativa has been established as hederagin 3-O-[O-α-L-arabinopyranosyl-(1 → 2)-β-D-glucopyranosyl-(1 → 2)-α-L-arabinopyranoside] 28-O-β-D-glucopyranoside. Compound (I), C52H84O22, mp 210–212°C, [α] 21D +38.4° (c 1.48; methanol). Acid hydrolysis of (I) led to hederogenin (II) — C30H48O4, mp 326–330°C, [α] 23D +84.2° (c 0.19; pyridine. The Hakomorimethylation of glycoside (I) yielded the permethylate (IV) — C65H11O22 [α] 23D +41.6° (c 1.79; methanol). The GLC analysis of the products of the methanolysis of compound (IV) showed the presence of 3,4,6-tri-O-methyl-D-glucopyranose, 2,3,4,6-tetra-O-methyl-D-glucopyranose, 3,4-di-O-methyl-L-O-arabinopyranose, and 2,3,4-tri-o-methyl-L-arabinopyranose. The alkaline hydrolysis of glycoside I gave compound (III) with mp 230–233°C, [α] 21D +35.2° (c 0.21; methanol), which was identified as medicoside C. Details of the PMR spectrum are given for compound (IV) and of the IR spectrum for compound (I).
Similar content being viewed by others
Literature cited
A. E. Timbekova and N. K. Abubakirov, Khim. Prir. Soedin., 451 (1984).
A. E. Timbekova and N. K. Abubakirov, Khim. Prir. Soedin., 805 (1985).
S. Hakomori, J. Biochem. (Tokyo),55, 205 (1964).
J. M. Van der Veen, J. Org. Chem.,28, 564 (1963).
W. Klyne, Biochem. J.,47, xli (1950).
Additional information
Institute of the Chemistry of Plant Substances of the Uzbek Academy of Sciences, Tashkent. Translated from Khimiya Prirodnykh Soedinenii, No. 5, pp. 607–610, September–October, 1986.
Rights and permissions
About this article
Cite this article
Timbekova, A.E., Abubakirov, N.K. Triterpene glycosides of alfalfa. III. Medicoside I. Chem Nat Compd 22, 571–574 (1986). https://doi.org/10.1007/BF00599263
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00599263