Abstract
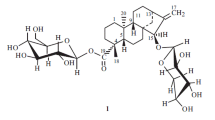
Two new triterpene glycosides — cucmariosides C1 and C2 — have been isolated from the Far Eastern holothurianEupentacta (=Cucumaria)fraudatrix Djakonov et Baranova. Their structures have been established with the aid of13C NMR and PMR spectroscopy, partial acid hydrolysis, periodate oxidation, and methylation as 16β-acetoxy-3-{[3-O-methyl-β-D-xylopyranosyl-(1→3)-β-D-glucopyranosyl-(1→4)] [β-D-xylopyranosyl-(1→2)]-β-D-quinovopyranosyl-(1→2)-β-D-xylopyranosyloxy}holosta-7,23,24(cis)-triene and 16β-acetoxy-3-{[3-O-methyl-β-D-xylopyranosyl-(1→3)-β-D-glucopyranosyl-(1→4)] [β-D-xylopyranosyl-(1→2)]-β-D-quinovopyranosyl-(1→2)-β-D-xylopyranosyloxy}holosta-7,22,24(trans)-triene, respectively.
Similar content being viewed by others
Literature cited
Sh. Sh. Afiyatullov, L. Ya. Tishchenko, V. A. Stonik, A. I. Kalinvoskii, and G. B. Elyakov, Khim. Prir. Soedin., 244 (1985).
J. D. Chanley and C. Rossi, Tetrahedron,25, 1897 (1969).
J. D. Chanley and C. Rossi, Tetrahedron,25, 1911 (1969).
A. S. Shashkov and O. S. Chizhov, Bioorg. Khim.,2, 437 (1976).
A. I. Kalinovskii, I. I. Mal'tsev, A. S. Antonov, and V. A. Stonik, Bioorg. Khim.,10, 1655 (1984).
Additional information
Pacific Ocean Institute of Bioorganic Chemistry of the Far-Eastern Scientific Center of the USSR Academy of Sciences, Vladivostok. Translated from Khimiya Prirodnykh Soedinenii, No. 6, pp. 831–837, November–December, 1987.
Rights and permissions
About this article
Cite this article
Afiyatullov, S.S., Kalinovskii, A.I. & Stonik, V.A. Structures of cucumariosides C1 and C2 — Two new triterpene glycosides from the holothurian Eupentacta fraudatrix. Chem Nat Compd 23, 691–696 (1987). https://doi.org/10.1007/BF00596644
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00596644