Abstract
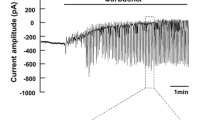
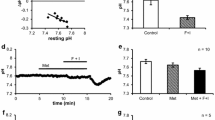
Cat submandibular glands were perfused with Locke solution in a thermostated chamber and intermittently stimulated with 10−5 M acetylcholine (ACh). In one series of experiments the perfusion pressure was varied within the range 90–60 mm Hg, and secretory flow rate, active K+-reuptake, passive K+-release, and resting and ACh-induced venous flow rates were measured. The ACh-induced secretory flow rate and the maximal K+-fluxes were related to the simultaneous ACh-induced venous flow rates. A proportionality was found between the maximal rate of ACh-induced K+-release and ACh-induced venous flow rates below 8 ml/min, while at higher flow rates the K+-release leveled off. The maximal rate of the post-stimulatory K+-reuptake increased proportionally to the ACh-induced perfusate flow rate throughout the range studied. The secretory flow rate was much less affected by changes in ACh-induced perfusate flow rate. In another series of experiments the gland temperature was varied within the range 12–37°C, and the same parameters were measured. All parameters decreased with cooling being reduced to 50% of their 37°C values at: 24°C for secretion, 19°C for K+-reuptake, and 14°C for K+-release. It is concluded: that 1) the rate of ACh-induced K+-release is limited by the ACh-induced perfusate flow rate (within the physiological range), 2) the capacity of the K+-reuptake mechanism is at least one order of magnitude larger than the maximal rate of K+-reuptake in vivo, 3) the marked temperature sensitivity of the secretory flow rate reflects the high complexity of the mechanisms involved.
Similar content being viewed by others
References
Biamino G, Wessel HJ (1973) Potassium induced relaxation of vascular smooth muscle: A possible mechanism of exercise hyperaemia. Pflügers Arch 343:95–106
Bloom SR, Edwards AV (1980) Vasoactive intestinal peptide in relation to atropine resistant vasodilatation in the submaxillary gland in the cat. J Physiol 300:41–53
Bundgaard M, Møller M, Poulsen JH (1977) Localization of sodium pump sites in cat salivary glands. J Physiol (Lond) 273:339–353
Burgen ASV (1956) The secretion of potassium in saliva. J Physiol (Lond) 132:20–39
Charnock JS, Doty DM, Russel JC (1971) The effect of temperature on the activity of (Na++K+)-ATPase. Arch Biochem Biophys 142:633–637
Creed KE, McDonald IR (1975) The effect of temperature and 2,4-dinitrophenol on transmembrane potentials of submandibular acinar cells. J Physiol (Lond) 247:521–535
Eveloff J, Karnaky KJ, Silva P, Epstein FH, Kinter WB (1979) Elasmobranch rectal gland cell. Autoradiographic localization of (3H)ouabain-sensitive Na, K-ATPase in rectal gland of dogfish,Squalus arcanthias. J Cell Biol 83:16–32
Folkow B, Fox RH, Krog J, Odelram H, Thorén O (1963) Studies on the reactions of the cutaneous vessels to cold exposure. Acta Physiol Scand 58:342–354
Hannafin E, Kinne-Saffran E, Friedman D, Kinne R (1983) Presence of a sodium-potassium chloride co-transport system in the rectal gland of squalus acanthias. J Membrane Biol 75:73–83
Kaladelfos G, Young JA (1972) Electrolyte secretion by the cat submaxillary gland stimulated with carbachol or isoproterenol. Proc Aust Physiol Pharmacol Soc 3:161
Laugesen LP, Nielsen JOD, Poulsen JH (1976) Partial dissociation between salivary secretion and active potassium transport in the perfused cat submandibular gland. Pflügers Arch 364:167–173
Laugesen LP, Nielsen JOD, Poulsen JH (1978) Dependence of salivary secretion, passive and active potassium transport on flow rate in the perfused submandibular gland. Acta Physiol Scand 102:19A-20A
Nauntofte B, Poulsen JH (1984) Chloride transport in the rat parotid acini: furosemide-sensitive uptake and calcium-dependent release. J Physiol 357:61P
Petersen OH, Maruyama Y (1984) Calcium-activated potassium channels and their role in secretion. Nature (Lond) 307:693–696
Petersen OH, Poulsen JH (1967) Inhibition of salivary secretion and secretory potentials by g-strophantin, dinitrophenol and cyanide. Acta Physiol Scand 71:194–202
Poulsen JH (1974) Effect of ouabain on two typs of active cation transport in the cat submandibular gland. In: Thorn NA, Petersen OH (eds) Secretory mechanisms of exocrine glands. Munksgaard, Copenhagen, pp 570–581
Poulsen JH (1975) Two phases of chorda-lingual induced vasodilatation in the cat's submandibular gland during prolonged perfusion with Locke solution. J Physiol (Lond) 253:79–94
Poulsen JH, Bledsoe SW (1978) Salivary gland K+ transport: in vivo studies with K+-specific microelectrodes. Am J Physiol 234:E79-E83
Poulsen JH, Kristensen LØ (1982) Is stimulation-induced uptake of sodium in the rat parotid acinar cells mediated by a sodium/chloride co-transport system? In: Case RM, Garner A, Turnberg LA, Young JA (eds) Electrolyte and water transport across gastrointestinal epithelia. Raven Press, New York, pp 199–208
Poulsen JH, Laugesen LP, Nielsen JOD (1982) Evidence supporting that basolaterally located Na+−K+-ATPase and a co-transport system for sodium and chloride are key elements in secretion of primary saliva. In: Case RM, Garner A, Turnberg LA, Young JA (eds) Electrolyte and water transport across gastrointestinal epithelia. Raven Press, New York, pp 157–159
Poulsen JH, Oakley B (1979) Intracellular potassium ion activity in resting and stimulated mouse pancreas and submandibular gland. Proc R Soc Lond B 204:99–108
Poulsen JH, Smaje LH, Ussing HH (1984) Evidence from O2 uptake measurements for (Na+−K+−2Cl−) co-transport in the rabbit submandibular gland. J Physiol (Lond) 348:43P
Silva P, Stoff J, Field M, Fine L, Forrest JN, Epstein FH (1977) Mechanism of active chloride secretion by shark rectal gland: Role of Na−K-ATPase in chloride transport. Am J Physiol 233:F298-F306
Smaje LH (1973) Spontaneous salivation in the rabbit submandibular gland. J Physiol (Lond) 231:179–193
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Dich-Nielsen, J.O., Laugesen, L.P. & Poulsen, J.H. Submandibular salivary secretion in the cat and associated potassium movements: Dependence on temperature and perfusate flow rate. Pflugers Arch. 403, 440–445 (1985). https://doi.org/10.1007/BF00589259
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00589259