Abstract
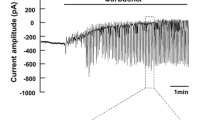
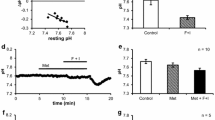
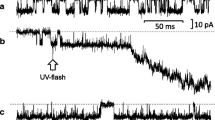
To assess the importance for transepithelial salt secretion of current flow across the baso-lateral membrane, we studied the effects of ouabain (1 mmol/l), Ba (3 mmol/l) and tetraethylammonium (TEA: 10 mmol/l) on secretion by the acinar (caerulein stimulated) and ductal (secretin stimulated) epithelia of the perfused rat pancreas. Within 10 min, ouabain caused a 79% inhibition of acinar secretion which was resolvable into two exponentials with half-times, respectively, of 0.24 min±0.19 (S.D.) and 6.40±0.46 min. In contrast, it caused only a monoexponential inhibition of ductal secretion (61% in 10 min) with a half-time of 5.08±0.26 min. Ba caused a monoexponential inhibition of acinar secretion (70% in 10 min) with a half-time of 1.82±0.27 min, but it had no inhibitory effect on ductal secretion. The action of TEA was similar to that of Ba: it caused monoexponential inhibition of acinar secretion (26% in 10 min) with a half-time of 1.82±0.03 min, and it too had no effect on ductal secretion. For comparison, we also studied the effect of these drugs on the more rapidly secreting rat mandibular gland (stimulated with acetylcholine). All three drugs were strongly inhibitory: within 10 min, ouabain caused a 95%, Ba an 89% and TEA an 83% inhibition. The decay curves appeared to be monoexponential with halftimes, respectively, of 1.49±0.12, 0.51±0.3 and 0.56±0.02 min. The results support the hypothesis that current flow across the baso-lateral membrane is an important concomitant of secretion by the pancreatic acinar epithelium and by mandibular glands, but they suggest that such a current is not important for secretion by pancreatic ducts.
Similar content being viewed by others
References
Allen DG, Eisner DA, Wray SC (1985) Sodium pump: birthday present. Nature 316:674–675
Bundgaard M, Møller M, Poulsen JH (1977) Localization of sodium pump sites in cat salivary glands. J Physiol (Lond) 273:339–353
Case RM, Conigrave AD, Novak I, Young JA (1980) Electrolyte and protein secretion by the perfused rabbit mandibular gland stimulated with acetylcholine or catecholamines. J Physiol (Lond) 300:467–487
Case RM, Hunter M, Novak I, Young JA (1984) The anionic basis of fluid secretion by the rabbit mandibular gland. J Physiol (Lond) 349:619–630
Findlay I, Petersen OH (1985) Acetylcholine stimulates a Ca2+-dependent Cl− conductance in mouse lacrimal acinar cells. Pflügers Arch 403:328–330
Fölsch UR, Creutzfeldt W (1977) Pancreatic duct cells in rats: secretory studies in response to secretin, cholecystokininpancreozymin, and gastrin in vivo. Gastroenterology 73:1053–1059
Greger R, Schlatter E (1984) Mechanism of NaCl secretion in rectal gland tubules of spiny dogfish (Squalus acanthias). II. Effect of inhibitors. Pflügers Arch 402:364–375
Greger R, Schlatter E, Gögelein H (1985) Cl−-channels in the apical cell membrane of the rectal gland “induced” by cAMP. Pflügers Arch 406:446–448
Hansen TR, Dineen DX, Petrak R (1984) Mechanism of action of barium ion on rat aortic smooth muscle. Am J Physiol 246:C235-C241
Higgins JT, Gebler B, Frömter E (1977) Electrical properties of amphibian urinary bladder epithelia. II. The cell potential profile inNecturus maculosus. Pflügers Arch 371:87–97
Iwatsuki N, Petersen OH (1985) Action of tetraethylammonium on calcium-activated potassium channels in pig pancreatic acinar cells studied by patch-clamp single-channel and whole-cell current recording. J Membr Biol 86:139–144
Iwatsuki N, Petersen OH (1985) Inhibition of Ca2+-activated K+ channels in pig pancreatic acinar cells by Ba2+, Ca2+, quinine and quinidine. Biochim Biophys Acta 819:249–257
Kempen HJM, De Pont JJHHM, Bonting SL (1977) Rat pancreas adenylate cyclase. V. Its preference in isolated rat pancreatic acinar cells. Biochim Biophys Acta 496:521–531
Klyce SD, Wong RK (1977) Site and mode of adrenaline action on chloride transport across the rabbit corneal epithelium. J Physiol (Lond) 266:777–799
Lingard JM, Young JA (1983) β-Adrenergic control of exocrine secretion by perfused rat pancreas in vitro. Am J Physiol 245:G690-G696
Machen TE, Forte JG (1984) Anion secretion by gastric mucosa. In: Gerencser GA (ed) Chloride transport coupling in biological membranes and epithelia. Elsevier, Amsterdam, pp 415–446
Martinez JR, Cassity N (1985)36Cl fluxes in dispersed rat submandibular acini: effects of acetylcholine and transport inhibitors. Pflügers Arch 403:50–54
Nagel W (1980) Time course of pump inhibition by ouabain in amphibian epithelia. Biochim Biophys Acta 599:736–740
Novak I, Davé C, Young JA (1984) The anionic basis of secretion by rat and rabbit mandibular glands. In: Case RM, Lingard JM, Young JA (eds) Secretion: Mechanisms and Control. Manchester University Press, Manchester, pp 77–80
Petersen OH, Maruyama Y (1984) Calcium-activated potassium channels and their role in secretion. Nature 307:693–696
Poulsen JH, Laugesen LP, Nielsen JOD (1982) Evidence supporting that basolaterally located Na+−K+-ATPase and a co-transport system for sodium and chloride are key elements in secretion of primary saliva. In: Case RM, Garner A, Turnberg LA, Young JA (eds) Electrolyte and water transport across gastrointestinal epithelia. Raven Press, New York, pp 157–159
Saito Y, Ozawa T, Hayashi H, Nishiyama A (1985) Acetylcholine-induced change in intracellular Cl−-activity of the mouse lacrimal acinar cells. Pflügers Arch 405:108–126
Schulz I, Ullrich KJ (1979) Transport processes in the exocrine pancreas. In: Giebisch G, Tosteson DC, Ussing HH (eds) Membrane Transport in Biology, Vol IVB, Transport Organs. Springer, Berlin, pp 811–852
Seow FKT, Lingard JM, Young JA (1986) The anionic basis of fluid secretion by rat pancreatic acini in vitro. Am J Physiol 250:G140-G148
Sewell WA, Young JA (1975) Secretion of electrolytes by the pancreas of the anaesthetized rat. J Physiol (Lond) 252:379–396
Silva P, Stoff J, Field M, Fine L, Forrest JN, Epstein FH (1977) Mechanism of active chloride secretion by shark rectal gland: role of Na-K-ATPase in chloride transport. Am J Physiol 233:F298-F306
Welsh MJ (1983) Inhibition of chloride secretion by furosemide in canine tracheal epithelium. J Membr Biol 71:219–226
Welsh MJ (1985) Basolateral membrane potassium conductance is independent of sodium pump activity and membrane voltage in canine tracheal epithelium. J Membr Biol 84:25–33
Welsh MJ, Smith PL, Frizzell RA (1983) Chloride secretion by canine tracheal epithelium. III. Membrane resistances and electromotive forces. J Membr Biol 71:209–218
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Evans, L.A.R., Pirani, D., Cook, D.I. et al. Intraepithelial current flow in rat pancreatic secretory epithelia. Pflugers Arch. 407 (Suppl 2), S107–S111 (1986). https://doi.org/10.1007/BF00584938
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00584938