Abstract
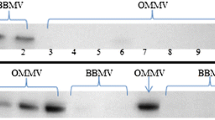
Thiol/disulfide is fundamental in protein function; we previously observed an inhibitory effect of thiol oxidants on the Na-dependent phosphate (Pi) uptake into renal brush border membrane vesicles (BBMV). We examined whether oxidation of glutathione (GSH) is involved in the mechanism. Vesicular thiols were measured by liquid chromatography. BBMV were incubated with reagents before an influx of Pi. Diamide (5 mM) reduced the capacity of the Pi uptake. Subsequent treatment with dithiothreitol (5 mM) blocked the inhibitory effect of diamide. Vesicular GSH was not modified only by the incubation, whereas it was oxidized by the treatment with diamide, and reduced by dithiothreitol. Furthermore, in vivo treatment with cAMP provided GSH-depleted BBMV without any influence on Pi uptake. Diamide did not inhibit the transport of Pi into GSH-depleted vesicles, but it did inhibit the uptake when GSH was introduced into the vesicles. In conclusion, a GSH-dependent mechanism is involved in the inhibitory effect of diamide on sodium-dependent Pi transport across the renal brush-border membrane.
Similar content being viewed by others
References
Bonjour, JP, Caverzasio J (1984) Phosphate transport in the kidney. Rev Physiol Biochem Pharmacol 100:161–214
Burckhardt G, Stern H, Murer H (1981) The influence of pH on phosphate transport into rat renal brush border membrane vesicles. Pflügers Arch 390:191–197
Chauez E, Brions R, Michel B, Braus C, Jay D (1985) Evidence for the involvement of dithiol groups in mitochondrial calcium transport: Studies with cadmium. Arch Biochem Biophys 242:493–497
Cheng L, Sacktor B (1981) Sodium gradient-dependent phosphate transport in renal brush border membrane vesicles. J Biol Chem 256:1556–1564
DeFranco AL, Koshland AL (1980) Multiple methylation in processing of sensory signals during bacterial chemotaxis. Proc Natl Acad Sci USA 77:2429–2433
Dousa TP, Kempson SA (1982) Regulation of renal brush border membrane transport of phosphate. Miner Electrolyte Metab 7:113–121
Frank PFH, Roelofsen P, Op den Kamp JAF (1982) Complete exchange of phosphatidylcholine from intact erythrocytes after protein crosslinking. Biochim Biophys Acta 687:105–108
Gilbert HF (1982) Biological disulfides: The third messenger? J Biol Chem 257:12086–12091
Gmaj P, Biber S, Stange G, Murer H (1984) Intravesicular NAD has no effect on sodium-dependent phosphate transport in isolated renal brush border membrane vesicles. Pflügers Arch 400:60–65
Hemmerman M, Hruska KA (1982) Cyclic AMP-dependent protein phospholylation in canine renal brush-border membrane vesicles is associated with decreased phosphate transport. J Biol Chem 257:992–999
Iriyama K, Iwamoto T, Yoshiura M (1986) Liquid chromatographic determination of glutathione with electrochemically pretreated glassy carbon electrode. J Liq Chromatogr 9:955–969
Isaacs J, Binkley F (1977) Glutathione dependent control of protein disulfide-sulfhydryl content by subcellular fractiona of hepatic tissue. Biochim Biophys Acta 497:192–204
Kuriyama R, Sakai H (1974) Role of tubulin-SH groups in polymerization to microtubules. Biochem J 76:651–654
May JM (1981) The role of glutathione in rat adipocyte pentose phosphate cycle activity. Arch Biochem Biophys 207:117–127
May JM (1982) The effect of insuline-stimulated pentose phosphate cycle activity on cellular glutathione content in rat adipocytes. Horm Metab Res 14:634–637
McIntyre T, Curthoys NP (1982) Renal catabolism of glutathione. J Biol Chem 257:11915–11921
Morgan EJ, Friedman E (1938) Maleic acid as inhibitor of enzyme reaction induced by SH-compounds. Biochem J 32:862–866
Reynolds R, Rea C, MacNamara PD, Segal S (1979) The effect of diamide on amino acid treatment by rat renal cortex slices. Biochim Biophys Acta 557:449–459
Shnellmann RG, Mandel LJ (1985) Intracellular comparmentation of glutathione in rabbit proximal tubules. Biochem Biophys Res Commun 133:1001–1005
Stein AF, Russell L, Klaassen CD (1986) High-performance liquid chromatographic analysis of glutathione and its thiol and disulfide degradation products. J Chromatogr 381:259–270
Suzuki M, Jo OD, Yanagawa N (1987) Possible role of thiol group and its redox state on phosphate uptake by rabbit renal brush border membrane vesicles. Kidney Int 31:358 (abstract)
Tate SS, Gran EM, Meister A (1979) Conversion of glutathione to glutathione disulfide by cell membrane-bound oxidase activity. Proc Natl Acad Sc USA 76:2710–2719
Turner RJ, George JN (1983) Evidence for two disulfide bonds important to the functioning of the renal outer cortical brush-border membraned-glucose transporter. J Biol Chem 258:3565–3570
Vanghn SP, Moss J (1981) Mono(ADP-ribosyl)transferases and their effects on cellular metabolism. Curr Top Cell Regul 20:205–246
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Suzuki, M., Iwamoto, T., Kawaguchi, Y. et al. Glutathione-dependent inactivation of sodium-dependent phosphate transport across rat renal brush-border membrane. Pflugers Arch. 413, 329–335 (1989). https://doi.org/10.1007/BF00584479
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00584479