Abstract
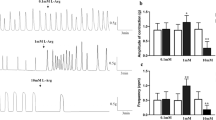
The effects of oxytocin and cAMP on the electrogenic Na+-transport in the short-circuited epithelium of the frog colon (Rana esculenta, Rana temporaria) were investigated. Oxytocin (100 mU · ml−1) elevated the shortcircuit current (I sc) transiently by 70% whereas cAMP (1 mmol · l−1) elicited a comparable sustained response. The mechanism of the natriferic action of cAMP was studied by analysing current fluctuations through apical Na+-channels induced by amiloride or CDPC (6-chloro-3,5-diaminopyrazine-2-carboxamid). The noise data were used to calculate Na+-channel density (M) and single apical Na+-current (i Na).i Na-Values obtained with amiloride and CDPC were 1.0±0.1 pA (n=5) and 1.1±0.2 pA (n=6) respectively and unaffected by cAMP. On the other hand, cAMP caused a significant increase in M from 0.23±0.08 μm−2 (n=5) to 0.49±0.17 μm−2 (n=5) in the amiloride experiments. In our studies with CDPC we obtained smaller values for M in control (0.12±0.04 μm−2;n=6) as well as during cAMP treatment (0.19±0.06 μm−2;n=6). However, the cAMP-induced increase in M was also significant. We conclude that cAMP stimulates Na+-transport across the frog colon by activating “silent” apical Na+-channels. Thus, the mechanism of regulation of colonic Na-transport in frogs differs considerably from that in other vertebrates as mammals and birds.
Similar content being viewed by others
References
Bridges RJ, Rummel W, Wollenberg P (1984) Effects of vasopressin on electrolyte transport across isolated colon from normal and dexamethasone-treated rats. J Physiol 355:11–23
Christensen O, Bindslev N (1982) Fluctuation analysis of short-circuit current in a warm-blooded sodium-retaining epithelium: site current, density, and interaction with triamterene. J Membr Biol 65:19–30
Clauss W, Dürr JE, Skadhauge E, Hörnicke H (1985) Effects of aldosterone and dexamethasone on apical membrane properties and Na-transport of rabbit distal colon in vitro. Pflügers Arch 403:186–192
Clauss W, Dürr JE, Guth D, Skadhauge E (1987) Effects of adrenal steroids on Na-transport in the lower intestine (coprodeum) of the hen. J Membr Biol 96:141–152
Cofré G, Crabbé J (1967) Active sodium transport by the colon of Bufo marinus: Stimulation by aldosterone and antidiuretic hormone. J Physiol 188:177–190
Cuthbert AW (1987) Comparative aspects of electrogenic sodium and electrogenic chloride transport in epithelial tissues. In: Kirsch R, Lahlou B (eds) Comparative physiology of environmental adaptations. Karger, Basel, pp 37–45
De Wolf I, Van Driessche W (1986) Voltage dependent Ba2+ block of K+ channels in apical membrane of frog skin. Am J Physiol 251:C696-C706
Eisenthal R, Cornish-Bowden A (1974) The direct linear plot. A new graphical procedure for estimating enzyme kinetic parameters. Biochem J 139:715–720
Erlij D, Van Driessche W, De Wolf I (1986) Oxytocin stimulates the apical K+ conductance in frog skin. Pflügers Arch 407: 602–606
Frizzell RA, Heintze K (1979) Electrogenic chloride secretion by the mammalian colon. In: Binder H (ed) Mechanisms of intestinal secretion. Liss, New York, pp 101–110
Helman SI, Cox TC, Van Driessche W (1983) Hormonal control of apical membrane Na transport in epithelia. Studies with fluctuation analysis. J Gen Physiol 82:201–220
Krattenmacher R, Clauss W (1988) Electrophysiological analysis of sodium-transport in the colon of the frog (Rana esculenta). Modulation of apical membrane properties by antidiuretic hormone. Pflügers Arch 411:606–612
Lewis SA (1983) Control of Na+ and water absorption across vertebrate “tight” epithelia by ADH and aldosterone. J Exp Biol 106:9–24
Li JH-J, Lindemann B (1983) Competive blocking of epithelial sodium channels by organic cations: The relationship between macroscopic and microscopic inhibition constants. J Membr Biol 76:235–251
Li JH-J, Palmer LG, Edelman IS, Lindemann B (1982) The role of sodium-channel density in the natriferic response of the toad urinary bladder to an antidiuretic hormone. J Membr Biol 64:77–89
Lindemann B (1984) Fluctuation analysis of sodium channels in epithelia. Annu Rev Physiol 46:497–515
Lindemann B, Van Driessche W (1977) Sodium-specific membrane channels of frog skin are pores: Current fluctuations reveal high turnover. Science 195:292–294
Macchia DD, Helman SI (1979) Transepithelial current-voltage relationships of toad urinary bladder and colon. Estimates ofE Na A and shunt resistance. Biophys J 27:371–392
Orloff J, Handler J (1967) The role of adenosine 3′,5′-phosphate in the action of antidiuretic hormone. Am J Med 42:757–768
Palmer LG, Li JH-J, Lindemann B, Edelman IS (1982) Aldosterone control of the density of sodium channels in the toad urinary bladder. J Membr Biol 64:91–102
Reif MC, Troutman SL, Schafer JA (1986) Sodium transport by rat cortical collecting tubule. Effects of vasopressin and desoxycorticosterone. J Clin Invest 77:1291–1298
Stetson DL, Lewis SA, Alles W, Wade JB (1982) Evaluation by capacitance measurements of antidiuretic hormone induced membrane area changes in toad bladder. Biochim Biophys Acta 689:267–274
Thompson L, Baxendale M, Helman SI (1987) Fluctuation analysis of ion transport by frog colon. Fed Proc 4:1269 (Abstract)
Van Driessche W, Erlij D (1983) Noise analysis of inward and outward Na+ currents across the apical border of ouabaintreated frog skin. Pflügers Arch 398:179–188
Van Driessche W, Gullentops K (1982) Conductance fluctuation analysis in epithelia. In: Baker PF (ed) Techniques in the life sciences. Techniques in cellular physiology, P133, County Clave, New York, Elsevier/North-Holland Scientific Publishers Ltd, pp 1–13
Van Driessche W, Lindemann B (1978) Low-noise amplification of voltage and current fluctuations arising in epithelia. Rev Sci Instrum 49:52–57
Van Driessche W, Lindemann B (1979) Concentration-dependence of currents through single sodium-selective pores in frog skin. Nature 282:519–520
Will PC, Cortright RN, DeLisle RC, Douglas JG, Hopfer U (1985) Regulation of amiloride-sensitive electrogenic sodium transport in the rat colon by steroid hormones. Am J Physiol 248:G124-G132
Wills NK, Zweifach A (1987) Recent advances in the characterization of epithelial ionic channels. Biochim Biophys Acta 906:1–31
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Krattenmacher, R., Fischer, H., van Driessche, W. et al. Noise analysis of cAMP-stimulated Na current in frog colon. Pflugers Arch. 412, 568–573 (1988). https://doi.org/10.1007/BF00583756
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00583756