Abstract
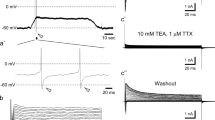
Motor nerve terminals in mouse and frog display behavior consistent with an appreciable permeability of the nerve terminal membrane to chloride. In mouse diaphragm, in the presence of 15 mM K+ and 2 mM or 8 mM Ca2+, replacement of Cl− by NO −3 , Br− or acetate causes a transient increase in the quantal release of acetylcholine, measured as the frequency of spontaneously occurring miniature end plate potentials (FMEPP); a rapid rise in FMEPP is followed by a slow decline, with a half-time of about 4 min, to an equilibration level close to the control level. After equilibration in a solution in which the Cl− is replaced by another anion, return to Cl−-containing solution causes a transient decrease in FMEPP with a subsequent slow recovery. The data are consistent with transient nerve terminal depolarization or hyperpolarization, reflecting a nerve terminal permeability to anions in the sequence Cl−>Br−>NO −3 >acetate. In 5 mM K+, changes in nerve terminal excitability, determined using focal stimulation, are also consistent with alteration of nerve terminal membrane potential as a consequence of anion substitution. The time course of relaxation of FMEPP after a change from Cl− to an anion of lower permeability, or vice versa, is considerably slower than that expected if Cl− permeability of nerve terminals is similar to that of skeletal muscle fibres, and if the nerve terminal behaves as a single compartment. In frog cutaneous pectoris, transient changes in FMEPP produced by substitution of anions in the bathing solution were similar to those produced in mouse diaphragm, but more rapid in time course.
Similar content being viewed by others
References
Aickin CC, Brading AF (1985) Advances in the understanding of transmembrane ionic gradients and permeabilities in smooth muscle obtained by using ion-selective micro-electrodes. Experimentia 41:879–887
Chiu SY, Ritchie JM (1982) Evidence for the presence of potassium channels in the internode of frog myelinated nerve fibres. J Physiol (Lond) 322:485–501
Cohen I, Van der Kloot W (1976) The effects of pH changes on the frequence of miniature end plate potentials at the frog neuromuscular junction. J Physiol (Lond) 262:401–414
Cole WV (1955) Motor ending in the striated muscle of vertebrates. J Comp Neurol 102:671–715
Cooke JD, Quastel DMJ (1973a)Transmitter release by mammalian motor nerve terminals in response to focal polarization. J Physiol (Lond) 228:377–405
Cooke JD, Quastel DMJ (1973b) The specific effect of potassium on transmitter release by motor nerve terminals and its inhibition by calcium. J Physiol (Lond) 228:435–458
Cooke JD, Okamoto K, Quastel DMJ (1973) The role of calcium in depolarization-secretion coupling at the motor nerve terminal. J Physiol (Lond) 228:459–497
Datyner MB, Gage PW (1980) Phasic secretion of acetylcholine at a mammalian neuromuscular junction. J Physiol (Lond) 303:299–314
Del Castillo J, Katz B (1954) Changes in endplate activity produced by presynaptic polarization. J Physiol (Lond) 124:586–604
Dreyer F, Peper K (1974) A monolayer preparation of innervated skeletal muscle fibres of the M. Cutaneus Pectoris of the frog. Pflügers Arch 248:257–262
Furshpan EJ (1956) The effects of osmotic pressure changes on the spontaneous activity at motor nerve endings. J Physiol (Lond) 134:689–697
Gage PW, Quastel DMJ (1966) Competition between sodium and calcium ions in transmitter release at mammalian neuromuscular junctions. J Physiol (Lond) 185:95–123
Hodgkin AL, Horowicz P (1959) The influence of potassium and chloride ions on the membrane potential of single muscle fibers. J Physiol (Lond) 148:127–160
Hutter OF, Padsha SM (1959) Effect of nitrate and other anions on the membrane resistance of frog skeletal muscle. J Physiol (Lond) 146:117–132
Landau EM (1969) The interaction of presynaptic polarization with calcium and magnesium in modifying spontaneous transmitter release from mammalian motor nerve terminals. J Physiol (Lond) 203:281–299
Liley AW (1956) The effects of presynaptic polarization on the spontaneous activity at the mammalian neuromuscular junction. J Physiol (Lond) 134:427–443
Noble D (1966) Application of Hodgkin-Huxley equations to excitable tissues. Physiol Rev 46:3–50
Palade PT, Barchi RL (1977) Characteristics of the chloride conductance in muscle fibers of the rat diphragm. J Gen Physiol 69:325–342
Quastel DMJ, Sastry BR, Steeves JD (1981) Focal excitation of motor nerve terminals. 8th Int Congr Pharmacol (IUPHAR), Abstract, p. 646
Rudel R, Lehmann-Horn F (1985) Membrane changes in cells from myotonia patients. Physiol Rev 65:310–356
Saint DA, Quastel DMJ (1986) Modification of motor nerve terminal excitability by alkanols and volatile anaesthetics. Brit J Pharmacol 88:747–756
Takeuchi A, Takeuchi N (1959) Active phase of frog's end-plate potential. J Neurophysiol 22:395–411
Weast RC (ed) (1969) Handbook of chemistry and physics50th edn., The Chemical Rubber Co.Cleveland, USA
Wright EM, Diamond JM (1977) Anion selectivity in biological systems. Physiol Rev 57:109–156
Author information
Authors and Affiliations
Additional information
This work was supported by grants from the Muscular DystrophyAssociation of Canada and the Medical Research Council of Canada
Rights and permissions
About this article
Cite this article
Saint, D.A., McLarnon, J.G. & Quastel, D.M.J. Anion permeability of motor nerve terminals. Pflugers Arch. 409, 258–264 (1987). https://doi.org/10.1007/BF00583474
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00583474