Abstract
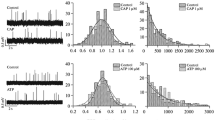
Pretreatment with hypertonic solutions, insulin, or adrenaline increases the size of quanta at the frog neuromuscular junction, as determined by measurements of miniature end plate potentials or currents (Van der Kloot and Van der Kloot 1985, 1986). The increase in quantal size apparently is due to an increase in acetylcholine (ACh) content of individual quanta. These treatments, therefore, can be used to study the packaging of ACh. Previously, I reported that increases are blocked by an inhibitor of active ACh uptake into vesicles (Van der Kloot 1986b, 1987b). The present study shows that the increases in quantal size were antagonized by inhibiting the Na+−K+ exchange pump with 100 μM ouabain, 10 μM dihydroouabain, or K+-free solutions. The increases in quantal size were also antagonized by 10 μM monensin, a Na+ ionophore, or by 5 μM aconitine, which opens Na+ channels at normal resting potentials. Apparently a rise in intracellular [Na+] inhibits the addition of ACh to quanta. The mechanism by which a rise in intracellular Na+ inhibits ACh packing is unknown, but apparently it is not due to inhibition of choline reuptake into the terminals.
Also consistent with the above hypothesis is that the increase in quantal size following depolarization for 2 h in elevated [K+]out was substantially enhanced when tetrodotoxin (TTX) was present, suggesting that in the absence of TTX there is a rise in [Na+]in that antagonizes the incorporation of additional ACh into the quanta.
Similar content being viewed by others
References
Anderson DC, King SC, Parsons SM (1983a) Pharmacological characterization of the acetylcholine transport system in purified torpedo electric organ synaptic vesicles. Mol Pharmacol 24:55–59
Anderson DC, King SC, Parsons SM (1983b) Inhibition of [H3]acetylcholine active transport by tetraphenyl borate and other anions. Mol Pharmacol 24:55–59
Attwell D, Cohen I, Eisner D, Ohba M, Ojeda C (1979) The steady-state TTX-sensitive (“window”) sodium current in cardiac Purkinje fibres. Pflügers Arch 379:137–142
Birks RI (1985) Activation of acetylcholine sythesis in cat sympathetic ganglia: dependence on external choline and sodium pump rate. J Physiol (Lond) 367:401–417
Birks RI, Cohen MW (1968) The action of sodium pump inhibitors on neuromuscular transmission. Proc R Soc Lond B Biol Sci 170:181–199
Clausen T (1986) Regulation of the active Na+−K+ transport in skeletal muscle. Physiol Rev 66:542–580
Cohen I, Van der Kloot W (1976) The effects of pH changes on the frequency of miniature end plate potentials at the frog neuromuscular junction. J Physiol (Lond) 262:401–414
Collier B, Ricny DM, Araujo DM (1986) Acetylcholine synthesis and release in a sympathetic ganglion in the presence of 2-(4-phenyl piperdino) cyclohexanol (AH5183). J Neurochem 46:822–830
Ducis I, Whittaker VP (1985) High-affinity, sodium-gradient-dependent transport of choline into vesiculated presynaptic plasma membrane fragments from the electric organ ofTorpedo marmorata and reconstitution of the solubilized transporter into liposomes. Biochim Biophys Acta 815:109–127
Elmqvist D, Quastel DMJ (1965) Presynaptic action of hemicholinium at the neuromuscular junction. J Physiol (Lond) 177:463–482
Fatt P, Katz B (1952) Spontaneous subthreshold activity at motor nerve endings. J Physiol (Lond) 117:109–128
Izumi F, Wada A, Yanagihara N, Kobayashi H, Toyohira Y (1986) Monensin-induced influx of22Na and the release of catecholamines in cultured bovine adrenal medulla cells. Biochem Pharmacol 35:2937–2940
Katz B (1969) The release of neural transmitter substances. Thomas, Springfield, Ill
Katz B, Thesleff S (1956) On the factors which determine the amplitude of the ‘miniature end-plate potential’. J Physiol (Lond) 138:267–278
Kita H, Van der Kloot W (1977) Time course and magnitude of changes in tonicity on acetylcholine release at frog neuromuscular junctions. J Neurophysiol 40:212–224
Koyano K, Kuba K, Minota S (1985) Long-term potentiation of transmitter release induced by repetitive presynaptic activities in bull-frog sympathetic ganglia. J Physiol 359:219–233
Marshall IG (1970) Studies on the blocking action of 2(4-phenyl-piperdino) cyclohexanol (AH5183). Br J Pharmacol 40:68–77
Marshall IG, Parsons RL (1975) The effects of tetraphenylboron on neuromuscular transmission in the frog. Br J Pharm 54: 325–332
Melega WP, Howard BD (1984) Biochemical evidence that vesicles are the source of the acetylcholine release from stimulated PC12 cells. Proc Natl Acad Sci USA 81:6536–6538
Schmidt H, Schmitt O (1974) Effect of aconitine on the sodium penetration of the node of Ranvier. Pflügers Arch 349:133–148
Schwartz A, Lindenmayer GE, Allen JC (1975) The sodium-potassium adenosine triphosphatase: pharmacological, physiological and biochemical aspects. Pharm Rev 27:3–134
Suchard SJ, Lattanzio FA Jr, Rubin RW, Pressman BC (1982) Stimulation of catecholamine secretion from cultured chromaffin cells by an ionophore-mediated rise in intracellular sodium. J Cell Biol 94:531–542
Toll L, Howard BD (1980) Evidence that an ATPase and a protonmotive force function in the transport of acetylcholine into storage vesicles. J Biol Chem 255:1787–1789
Tucek S (1983) The synthesis of acetylcholine. In: Lajtha A (ed) Handbook of neurochemistry. Plenum Press, New York, pp 219–249
Van der Kloot W (1986a) 2-(4-Phenylpiperidino) cyclohexanol (AH5183) decreases quantal size at the frog neuromuscular junction. Pflügers Arch 406:83–85
Van der Kloot W (1986b) AH5183 blocks increases in quantal size at the frog neuromuscular junction. J Physiol (Lond) 373:66P
Van der Kloot W (1986c) Insulin-induced increases in the amount of acetylcholine/quanta are blocked by Na+−K+ pump inhibition. Regul Pept 15:194
Van der Kloot W (1987a) Pretreatment with hypertonic solutions increases quantal size at the frog neuromuscular junction. J Neurophysiol 57:1536–1554
Van der Kloot W (1987b) Ammonium inhibits the packing of acetylcholine into quanta. FASEB J 1:298–302
Van der Kloot W (1988) Evidence for two components of release in quanta from isolated pretreated frog neuromuscular junctions. J Physiol (Lond) (in press)
Van der Kloot W, Cohen IS (1984) Temperature effects on spontaneous and evoked release at the frog neuromuscular junction. J Neurosci 4:2200–2203
Van der Kloot TE, Van der Kloot W (1983) Increasing quantal size at the frog neuromuscular junction. J Physiol (Lond) 349:51P
Van der Kloot W, Van der Kloot TE (1985) Activity increases quantal size at the frog neuromuscular junction. Experientia 41:47–48
Van der Kloot W, Van der Kloot TE (1986) Catecholamines, insulin and ACTH increase quantal size at the frog neuromuscular junction. Brain Res 376:378–381
Vizi ES, Vyskocil F (1979) Changes in total and quantal release of acetylcholine in the mouse diaphragm during activation and inhibition of membrane ATPase. J Physiol (Lond) 286:1–14
Vyas S, Marchbanks RM (1981) The effect of ouabain on the release of [14C]acetylcholine and other substances from synaptosomes. J Neurochem 37:1467–1474
Whitton PS, Marshall IG, Parsons SM (1986) Reduction of quantal size by vesamicol (AH5183), and inhibitor of vesicular acetylcholine storage. Brain Res 385:189–192
Winkler H, Apps DK, Fischer-Colbrie R (1986) The molecular function of adrenal chromaffin granules: established facts and unresolved topics. Neuroscience 18:261–290
Yamamura H, Snyder SH (1972) Choline: high affinity uptake by rat brain synaptosomes. Science 178:626–628
Yu SP, Van der Kloot W (1987) The increase in quantal size produced by hypertonic solutions and it's antagonism by AH5183 and Na+−K+ pump inhibition at mouse neuromuscular junctions. Fed Proc 46:337
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Van der Kloot, W. The packing of acetylcholine into quanta at the frog neuromuscular junction is inhibited by increases in intracellular sodium. Pflugers Arch. 412, 258–263 (1988). https://doi.org/10.1007/BF00582506
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00582506