Abstract
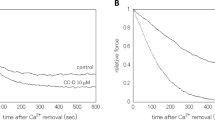
The effects of pH (from pH values 6.50–7.10) on isometric tension development and relaxation were investigated in Triton X-100 “skinned” rat caudal artery. Helically cut skinned strips contracted in 21 μM Ca2+ were studied with respect to maximal isometric tension (Po) and rate of contraction (T0.5C), and following relaxation in 18 nm Ca2+, the rate of relaxation (T0.5R). Acidic pH (pH 6.50) decreased Po to 87% of isometric force obtained at pH 6.90, and increased the rate of contraction as shown by a decrease of T0.5C to 80%. In contrast, T0.5R increased 4.5-fold, indicating that with a change of only 0.40 pH units, relaxation rates were dramatically decreased. pCa-tension curves at pH values 6.50, 6.70, 6.90 and 7.10 indicated no significant shift in half maximal activation (pCa50) between pH 6.50 and 6.70, but a significant (P<0.01) shift in pCa50 between pH 6.70 ([Ca2+]=0.46 μM) and pH 7.10 ([Ca2+]=0.87 μM). Compared to contractions at pH 6.90, myosin light chain (LC20) phosphorylation at pH 6.50 was significantly greater at 30 and 60 s into contraction but not significantly different at 3–10 min. At both pH 6.50 and 6.90, dephosphorylation was rapid and substantially preceded relaxation; LC20 dephosphorylation and relaxation occurred more rapidly at pH 6.90 than at 6.50. At pH 6.50 and 6.90, relax solutions made with increased Ca2+ buffering capacity showed no effect in enhancing T0.5R, suggesting the difference between relaxation rates was not due to Ca2+ diffusion limitations from the skinned strip. We suggest pH changes can after the contractile and relaxation responses in vascular smooth muscle and these effects may be related to LC20 phosphorylation/dephosphorylation regulatory mechanisms.
Similar content being viewed by others
Abbreviations
- PIPES:
-
piperazine-N,N′-bis-(2-ethanesulfonic acid)
- EGTA:
-
5-ethyleneglycol-bis-(β-aminoethyl ether)-N,N,N′,N′-tetraacetic acid
- DTT:
-
dithiothreitol
- HDTA:
-
1,6-diaminohexane-N,N,N′,N′-tetraacetic acid
- SDS:
-
sodium dodecyl sulfate
References
Adelstein RS, Klee CB (1981) Purification and characterization of smooth muscle myosin light chain kinase. J Biol Chem 256: 7501–7509
Aickin CC (1984) Direct measurement of intracellular pH and buffering power in smooth muscle cells of guinea-pig vas deferens. J Physiol (Lond) 349:571–585
Bialojan C, Merkel L, Rüegg JC, Gifford D, Di Salvo J (1985) Prolonged relaxation of detergent-skinned smooth muscle involves decreased endogenous phosphatase activity. Proc Soc Exp Biol Med 178:648–652
Blumenthal DK, Stull JT (1982) Effects of pH, ionic strength, and temperature on activation by calmodulin and catalytic activity of myosin light chain kinase. Biochemistry 21:2386–2391
Chatterjee M, Murphy RA (1983) Calcium-dependent stress maintenance without myosin phosphorylation in skinned smooth muscle. Science 221:464–466
Diecke FPJ, Gardner J, Hausser R (1986) Mathematical modelling of the simultaneous diffusion of EGTA, CaEGTA, and Ca2+ in a two-compartmental system representing skinned smooth muscle. Biophys J 49:73a (abstr)
Dillon PF, Aksoy MO, Driska SP, Murphy RA (1981) Myosin phosphorylation and the cross-bridge cycle in arterial smooth muscle. Science 211:495–497
Driska S, hartshorne DJ (1975) The contractile properties of smooth muscle. Properties and components of a Ca2+-sensitive actomyosin from chicken gizzard. Arch Biochem Biophys 167:203–212
Driska SP, Aksoy MO, Murphy RA (1981) Myosin light chain phosphorylation associated with contraction in arterial smooth muscle. Am J Physiol 240:C222-C233
Fisher MJ, Dillon PF (1986) Graded metabolic impairment of smooth muscle. Biophys J 49:101a (abstr)
Gardner JP (1984) Relations among myosin light chain phosphorylation, Ca2+ and isometric force in skinned porcine carotid artery. Ph. D. thesis dissertation, University of Cincinnati University Microfilms Int., Ann Arbor, MI
Gordon AR (1978) Contraction of detergent-treated smooth muscle. Proc Natl Acad Sci USA 75:3527–3530
Hellstrand P, Vogel HJ (1985) Phosphagens and intracellular pH in intact rabbit smooth muscle studied by31P-NMR. Am J Physiol 248:C320-C329
Herlihy JT (1980) Helically cut vascular strip preparation: geometrical considerations. Am J Physiol 238:H107-H109
Hoar PE, Kerrick WGL, Cassidy PS (1979) Chicken gizzard: relation between calcium-activated phosphorylation and contraction. Science 204:503–506
Hoar PE, Pato MD, Kerrick WGL (1985) Myosin light chain phosphatase: effect on the activation and relaxation of gizzard smooth muscle skinned fibers. J Biol Chem 260:8760–8764
Hoar PE, Kenney RE, Kerrick WGL (1986) Mg2+ affects maximum tension and relaxation rates in skinned smooth muscle cells, but not the pCa-tension relationship. Biophys J 51:332a (abstr)
Iino M (1981) Tension responses of chemically skinned fibre bundles of the guinea-pig taenia caeci under varied ionic environments. J Physiol (Lond) 320:459–467
Kamm KE, Stull JT (1985a) The function of myosin and myosin light chain kinase phosphorylation in smooth muscle. Annu Rev Pharmacol Toxicol 25:593–620
Kamm KE, Stull JT (1985b) Myosin phosphorylation, force and maximal shortening velocity in neurally stimulated tracheal smooth muscle. Am J Physiol 249:C238-C247
Kato S, Ogasawara T, Osa T (1982) Calcium diffusion in uterine smooth muscle sheets. J Gen Physiol 80:257–277
Kato S, Osa T, Ogasawara T (1984) Kinetic model for isometric contraction in smooth muscle on the basis of myosin phosphorylation hypothesis. Biophys J 46:35–44
Keller CH, Olwin BB, La Porte DC, Storm DR (1982) Determination of the free energy coupling for binding of calcium ions and troponin I to calmodulin. Biochemistry 21:156–162
Kulbertus H (1964) Etude des échanges d'ions H+ au cours de la contraction des parios carotidiennes soumises à différents agents vasomoteurs. Angiologica 1:275–278
Marston SB (1982) The regulation of smooth muscle contractile proteins. Prog Biophys Mol Biol 41:1–41
Moisescu DG (1976) Kinetics of reaction in calcium-activated skinned muscle fibres. Nature 262:610–613
Mrwa U, Hartshorne DJ (1980) Phosphorylation of smooth muscle myosin and myosin light chains. Fed Proc 39:1564–1568
Mrwa U, Achtig I, Rüegg JC (1974) Influences of calcium concentration and pH on the tension development and ATPase activity of the arterial actomyosin contractile system. Blood Vessels 11:277–286
Murphy RA (1971) Arterial actomyosin: effects of pH and temperature on solubility and ATPase activity. Am J Physiol 220: 1494–1500
Murphy RA (1980) Mechanics of vascular smooth muscle. In: Bohr DF, Somlyo AP, Sparks HV, Jr (eds) Handbook of physiology — vascular smooth muscle, vol 2. American Physiological Society, Bethesda, MD, pp 325–351
Murphy RA, Aksoy MO, Dillon PF, Gerthoffer WT, Kamm KE (1983) The role of myosin light chain phosphorylation in regulation of the crossbridge cycle. Fed Proc 42:51–56
Nakayama S, Tomita T, Seo Y, Watari H (1986) P-31 NMR studies in smooth muscles of guinea-pig stomach and taenia coli. Proc Int Union Physiol Sci XVI:67(abstr)
Oakley BR, Kirsch DR, Morris NR (1980) A simple ultrasensitive silver stain for detecting proteins in polyacrylamide gels. Anal Biochem 98:231–237
Ogawa Y, Tanokura M (1984) Calcium binding to calmodulin: effects of ionic strength, Mg2+, pH and temperature. J Biochem 95:19–28
Pato MD, Adelstein RS (1983) Characterization of a Mg2+-dependent phosphatase from turkey gizzard smooth muscle. J Biol Chem 258:7055–7058
Perrin DD, Sayce IG (1967) Computer calculation of equilibrium concentrations in mixtures of metal ions and complexing species. Talanta 14:833–842
Peterson JW 3d (1982) Rate-limiting steps in the tension development of freeze-glycerinated vascular smooth muscle. J Gen Physiol 79:437–452
Schädler M (1967) Proportionale Aktivierung von ATPase-Aktivität und Kontraktions-Spannung durch Calciumionen in isolierten contractilen Strukturen verschiedener Muskelarten. Pflügers Arch 296:70–90
Schneider M, Sparrow M, Rüegg JC (1981) Inorganic phosphate promotes relaxation of chemically skinned smooth muscle of guinea-pig Taenia coli. Experientia 37:980–982
Siegman MJ, Butler TM, Mooers SU, Michalek A (1984) Ca2+ can affectV max without changes in myosin light chain phosphorylation in smooth muscle. Pflügers Arch 401:385–390
Sillen LG, Martell AE (1971) Stability constants of metal-ion complexes. Suppl No 1. Burlington House, London, pp 731–732
Sobieszek A (1987) MgATPase activity of vertebrate smooth muscle actomyosin: stimulation by tropomyosin is modified by myosin phosphorylation and its conformational state. In: Siegman MJ, Somlyo AP, Stephens NL (eds) Progress in clinical and biological research: regulation and contraction of smooth muscle, vol 245. Liss, New York, pp 159–181
Stephens NL, Kroeger EA, Low W (1977) Intracellular pH in hypoxic smooth muscle. Am J Physiol 232:E330-E335
Stout MA, Diecke FPJ (1983)45Ca sequestration and release in chemically skinned strips of vascular smooth muscle. J Pharmacol Exp Ther 225:102–111
Vermuë NA, Nicolay K (1983) Energetics of smooth muscle taenia caecum of guinea-pig: a31P-NMR study. FEBS Lett 156: 293–297
Author information
Authors and Affiliations
Additional information
J. P. Gardner was supported in part by an AHA N.J. affiliate postdoctoral fellowship
Rights and permissions
About this article
Cite this article
Gardner, J.P., Diecke, F.P.J. Influence of pH on isometric force development and relaxation in skinned vascular smooth muscle. Pflugers Arch. 412, 231–239 (1988). https://doi.org/10.1007/BF00582502
Received:
Revised:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00582502