Abstract
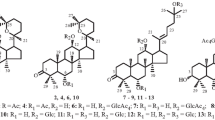
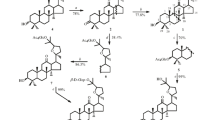
The glycosylation of 20(S),24(R)-epoxydammarane-3,12β,25-triols under the conditions of the previous formation of an ion pair with a Lewis acid and subsequent treatment with cholesteryl (α-D-glucose orthoacetate) leads to the selective formation with high yields of the corresponding 12-monoglucosides having the trans configuration of the glucosidic bond. The regioselectivity of the direct glycosylation of 20(S), 24(R)-epoxydammarane-3,12β,25-triols by orthoesters is determined by the influence of intramolecular hydrogen bonds in the initial triols. Details of the PMR and13C NMR spectra of the new compounds obtained are given.
Similar content being viewed by others
Literature cited
L. N. Atopkina and N. I. Uvarova, Khim. Prir. Soedin., 329 (1981).
N. F. Samoshina, L. N. Atopkina, V. L. Novikov, V. A. Denisenko, and N. I. Uvarova, Khim. Prir. Soedin., 596 (1982).
H. Beierbeck, J. K. Saunders, and J. W. Apsimon, Can. J. Chem.,55, 2813 (1977).
O. Tanaka and S. Yahara, Phytochemistry,17, 1353 (1978).
V. F. Samoshina, V. L. Novikov, V. A. Denisenko, and N. I. Uvarova, Khim. Prir. Soedin., 185 (1983).
N. K. Kochetkov, A. Ya. Khorlin, and A. F. Bochkov, Tetrahedron,23, 693 (1967).
Additional information
Pacific Ocean Institute of Bioorganic Chemistry, Far Eastern Scientific Center, Academy of Sciences of the USSR, Vladivostok. Translated from Khimiya Prirodnykh Soedinenii, No. 3, pp. 315–321, May–June, 1983.
Rights and permissions
About this article
Cite this article
Samoshina, N.F., Novikov, V.L., Denisenko, V.A. et al. Regio- and stereoselective glycosylation of 20(S), 24(R)-epoxydammarane-3,12β,25-triols with cholesteryl (α-D-glucose orthoacetate). III. Chem Nat Compd 19, 299–304 (1983). https://doi.org/10.1007/BF00579764
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00579764