Abstract
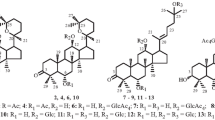
The rearrangement of pregnenolone and 16-dehydropregnenolone α-D-glucose orthoacetates in the presence of mercuric bromide is, because of the high specific selectivity and satisfactory yields of the desired β-D-glucosides, the most effective method of glycosylating the steroids mentioned.
Similar content being viewed by others
Literature cited
B. Pal and M. Gyorgy, Magy. Kem. Folyoirat,72, 201 (1966).
R. Kocovsky, K. K. Kosoev, and Z. Proshazka, Collect. Czech. Chem. Commun.,38, 3273 (1973).
N. I. Uvarova, G. I. Oshitok, and G. B. Elyakov, Carbohydr. Res.,27, 79 (1973).
M. Mazurek and A. S. Perlin, Can. J. Chem.,43, 1918 (1965).
N. F. Samoshina and N. I. Uvarova, Khim. Prir. Soedin., 334 (1979).
H. Eggert, C. L. Van Antwerp, N. S. Bhacca, and C. Djerassi, J. Org. Chem.,41, 71 (1976).
J. W. Blunt and J. B. Stothers, Org. Magn. Reson.,9, 439 (1977).
N. K. Kochetkov, A. Ya. Khorlin, and A. F. Bochkov, Tetrahedron,23, 693 (1967).
G. Wulf, and W. Schmidt, Carbohydr. Res.,53, 33 (1977).
N. I. Uvarova, N. F. Samoshina, G. I. Oshitok, and G. B. Elyakov, Izv. Akad. Nauk SSSR, Ser. Khim., 1907 (1975).
L. Fieser and M. Fieser, Steroids, Reinhold, New York (1959).
Additional information
Pacific Ocean Institute of Bioorganic Chemistry, Far Eastern Scientific Center, Academy of Sciences of the USSR, Vladivostok. Translated from Khimiya Prirodnykh Soedinenii, No. 2, pp. 191–196, March–April, 1984.
Rights and permissions
About this article
Cite this article
Samoshina, N.F., Denisenko, V.A., Novikov, V.L. et al. Catalytic rearrangement ofα-D-glucose 1,2-orthoacetate derivatives of pregnenolone and 16-dehydropregnenolone. Chem Nat Compd 20, 177–182 (1984). https://doi.org/10.1007/BF00579479
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00579479