Abstract
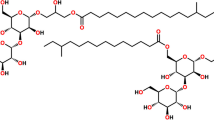
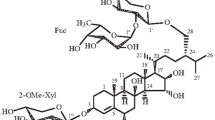
From an ethanolic extract of the holothuriansStichopus chloronotus by column chromatography on silica gel a new triterpene oligoside has been isolated the structure of which has been established as 23(S)-acetoxy-3β-{4′-O-[O-(3-O-methyl-β-D-glucopyranosyl)-(1→3)-β-D-glucopyranosyl]-2′-O-[O-(3-O-methyl-β-D-glucopyranosyl(-(1→3)-O-β-D-xylopyranosyl-(1→4)-β-D-glucopyranosyl]-β-D-xylopyranosyloxy}holost-7-ene. A hypothesis has been put forward concerning the biosynthesis of the carbohydrate chains in the glycosides of holothurians of the orderAspidochirota from bioside blocks.
Similar content being viewed by others
Literature cited
V. A. Stonik, I. I. Mal'tsev, A. I. Kalinovskii, C. Conde, and G. B. Elyakov, Khim. Prir. Soedin., 194 (1982).
A. I. Kalinovskii, V. F. Sharypov, V. A. Stonik, A. K. Dzizenko, and G. B. Elyakov, Bioorg. Khim.,6, 86 (1980).
G. B. Elyakov, V. A. Stonik, et al., Dokl. Akad. Nauk SSSR, 259, No. 6, 1367 (1981).
A. S. Shashkov and O. S. Chizhov, Bioorg. Khim.,2, 437 (1976).
J. K. Hamilton and F. Smith, J. Am. Chem. Soc.,78, 5907 (1955).
S. Hakomori, J. Biochem. (Tokyo),55, 205 (1964).
T. Usui, J. Chem. Soc., 2425 (1974).
V. F. Sharypov, A. D. Chumak, V. A. Stonik, and G. B. Elyakov, Khim. Prir. Soedin., 181 (1981).
I. Kitagawa, M. Kobayashi, M. Nori, and Y. Kyogoku, Chem. Pharm. Bull.,29, 282 (1981).
I. Kitagawa, T. Nishino, and Y. Kyogoku, Tetrahedron Lett., 985 (1978).
I. Kitagawa, T. Inamoto, M. Fushida, S. Okada, M. Kobayashi, T. Nishino, and Y. Kyogoky, Chem. Pharm. Bull.,28, 1651 (1980).
I. Kitagawa, T. Nishino, T. Matsuno, H. Akutsu, and Y. Kyogoku, Tetrahedron Lett., 1418 (1979).
I. Kitagawa, H. Yamanaka, M. Kobayashi, T. Nishino, I. Yosioka, and T. Sugawara, Chem. Pharm. Bull.,26, 3722 (1978).
A. Kelecom, B. Tursch, and M. Vanhaelen, Bull. Soc. Chim. Belg.,85, 277 (1976).
Additional information
Pacific Ocean Institute of Bioorganic Chemistry, Far Eastern Scientific Center, Academy of Sciences of the USSR, Vladivostok. Translated from Khimiya Prirodnykh Soedinenii, No. 2, pp. 200–204, March–April, 1982.
Rights and permissions
About this article
Cite this article
Stonik, V.A., Mal'tsev, I.I., Kalinovskii, A.I. et al. Glycosides of marine invertebrates. XII. Structure of a new triterpene oligoglycoside from holothurians of familyStichopodidae . Chem Nat Compd 18, 182–186 (1982). https://doi.org/10.1007/BF00577189
Received:
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00577189