Abstract
Induction of inhibited growth halo, around filter paper discs or groundnut seeds dusted with aldrex T, diameter of which consistently increased with dressing application rate, was exhibited by the root nodule bacteriumRhizobium leguminosarum. Some cells sensitive to aldrex T gained recovery upon transference to fresh media denoting a bacteriostatic effect of the anti-fungal dressing.
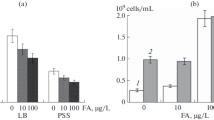
The viable bacterial cells steadily declined with fungicide concentrations between 100 and 500 p.p.m. An increase of 130% in viable colony count was recorded when the dose was increased to 700 p.p.m., and a relatively similar increase at 900 p.p.m., thus exhibiting a zone of inverted growth at 500 p.p.m. which was also confirmed by the optical density test. The latter was reliable after 24h incubation when the turbidity uniformly deminished with the fungicide dose to a maximal decline at 500 p.p.m. Aldrex T decreased the log and increased the stationary phases ofR. leguminosarum growth at all test doses.
In vitro treatment ofR. leguminosarum with the fungicide exhibited some changes in bacterial morphology, cell lysis and shrinkage of cell membrane as revealed by the electron microscope.
Similar content being viewed by others
References
Abdalla, M. H. 1975. Growth responses of two phytopathogenic fungi to fernasan in culture media. Mycopathologia 55: 169–173.
Abdalla, M. H. & S. M. Suleiman. 1978. Interference of seed dressing on groundnut nodulation Mycopathologia (in press).
Alexander, M. 1961. Introduction to soil microbiology. John Wiley & Sons, IMC. New York, London & Sydney.
Ballen, W. B. 1961. Interaction between pesticides and soil micro-organisms. Ann. Rev. Microbiol. 15: 69.
Buchanan, R. E. & N. E. Gibbons. 1974. Bergey's manual of determinative bacteriology, p. 543. The Williams and Wilkan Company.
Crosier, W. F. & W. E. Walls. 1968. Biological determination of the amount of mercury on seeds. Proc. Assoc. Official Seed Analysts 58: 37. (cited by H. Ehle, 1973).
Dirar, H. 1977. Personal communication.
Ehle, H. 1973. Microbiological methods for detecting dressings on treated seeds. In: Residual Review 45: 125–145 (ed. F. A. Gunther, S. Verlag, C. V. Clayton & D. E. Ellis, New York & Heidelberg-Berlin).
Fisher, D. J. 1976. Effect of pesticides on Rhizobium trifolii and its symbiotic relationship with white clover. Pest. Sci. 5: 219–222.
Hamdi, Y. A. 1976. Yield and green-house experiments on the response of legumes in Egypt to inoculation and fertilizers. In: Symbiotic nitrogen fixation in plants (ed. P. S. Nutman, Cambridge University Press).
Horsfall, J. G. & A. E. Dimond. 1960. Plant pathology, vol. II, pp. 527–533. Academic Press, New York & London.
Leben, C. & G. W. Keitt. 1950. A bioassay for tetramethyl thiram disulphide. Phytopathol. 40: 950.
Machacek, J. E. 1950. An agar-sheet method of testing the efficiency of seed treating machines. Can. J. Research Sect. C28: 739.
Newman, A. S. & C. E. Downing. 1948. The physiology and biochemistry of herbicides. Academic Press, London.
Pareek, R. P. & A. C. Gour. 1970. Effect of dichlorodiphenyl trichloroethane (DDT) on symbiosis of Rhizobium spp. with Phaseolus aureus. Plant and Soil 33: 297–304.
Pepper, E. H. & K. A. Claussen. 1963. A rapid bio-assay for the detection of seed pesticides. Plant Disease Reporter 47: 374.
Targeson, D. C. 1967. Fungicides: Agriculture and industrial applications and environmental interactions. Academic Press, New York.
Vincint, J. H. 1970. A manual for the practical study of the root nodule bacteria. Blackwell Scientific Publication, Oxford & Edinburgh.
Wash, D.C. 1949. Pest control, burent and plant quarantine. (From: Chemical Abstracts 44, 2690).
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Abdalla, M.H., Suleiman, S.M. Sensitivity of the root nodule rhizobium leguminosarum to the anti-fungal dressing aldrex T. Mycopathologia 64, 137–142 (1978). https://doi.org/10.1007/BF00576363
Issue Date:
DOI: https://doi.org/10.1007/BF00576363