Summary
-
1.
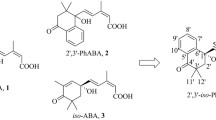
A number of aryl analogs of abscissic acid have been obtained by the condensation of substituted benzylideneacetones with ethyl bromoacetate by the Reformatsky reaction. Furyl and other heterocyclic analogs of abscissic acid have been synthesized for the first time by the Wittig reaction of substituted furfurylideneacetones and ketones containing heterocyclic nuclei with ethoxycarbonylmethylenetriphenylphosphorane.
-
2.
It has been found by PMR spectroscopy and also by GLC analysis that the Reformatsky condensation leads predominantly to the formation of the trans,trans isomer in the mixture. The Wittig reaction gives the cis,trans isomer in predominating amount.
-
3.
Biotests have shown that the activities of the analogs synthesized are inferior to the activity of abscissic acid itself.
Similar content being viewed by others
Literature cited
N. D. Livshits, Ch. Sh. Kadyrov, and V. I. Kefeli, Khim. Prirodn. Soedin., 73 (1970).
N. D. Livshits, N. D. Abdullaev, and Ch. Sh. Kadyrov, Uzb. Khim. Zh., No. 2, 42 (1976).
N. S. Vul'fson and L. Kh. Vinograd, Reactions and Methods of Investigating Organic Compounds [in Russian], Vol. 17, Moscow (1967).
L. Kh. Vinograd and N. S. Vul'fson, Dokl. Akad. Nauk SSSR,123, 97 (1958).
A. V. El'tsov, Zh. Obshch. Khim.,32, 1525 (1962).
H. R. Hensel, Chem. Ber.,4, 1325 (1965).
Preparative Organic Chemistry [in Russian], Moscow (1964), p. 554.
J. D. Cawley and D. R. Nelan, J. Am. Chem. Soc.,77, 4130 (1955).
Additional information
Institute of the Chemistry of Plant Substances, Academy of Sciences of the Uzbek SSR. Tashkent. Translated from Khimiya Prirodnykh Soedinenii, No. 1, pp. 63–70, January–February, 1978.
Rights and permissions
About this article
Cite this article
Livshits, N.D., Kadyrov, C.S. & Abdullaev, N.D. Synthesis of aromatic and heterocyclic analogs of the natural growth inhibitor abscissic acid. Chem Nat Compd 14, 49–54 (1978). https://doi.org/10.1007/BF00565875
Received:
Issue Date:
DOI: https://doi.org/10.1007/BF00565875