Conclusions
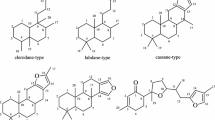
It has been shown that the dehydration of the sesquiterpene lactone coronopilin with SOCl2 forms Δ1(2)-anhydrocoronopilin. The configuration of the methyl groups at C5 and C10 in anhydrocoronopilin has been established by analogy with the conversion products of the pseudoguaianolide parthenin.
Similar content being viewed by others
References
V. I. Novikov, Yu. N. Forostyan, and D. P. Popa, KhPS [Chemistry of Natural Compounds], 5, no. 6, 1969.
W. Herz, H. Watanabe, M. Miyazaki, and Y. Kishida, J. Am. Chem. Soc., 13, 2601, 1962.
Author information
Authors and Affiliations
Additional information
Khimiya Prirodnykh Soedinenii, Vol. 6, No. 1, pp. 29–31, 1970.
Rights and permissions
About this article
Cite this article
Novikov, V.I., Forostyan, F.N. & Popa, D.P. A sesquiterpene lactone ofCyclachaena xanthifolia-anhydrocoronopilin. Chem Nat Compd 6, 26–27 (1970). https://doi.org/10.1007/BF00564151
Published:
Issue Date:
DOI: https://doi.org/10.1007/BF00564151