Summary
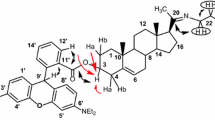
1. The structure of norechinocystenolone has been established and it has been shown to be identical with the natural nortriterpene albigenin.
2. The isomerization of isonorechinocytenedione into norechinocystenedione consists in the migration of a double bond from 12–13 position to the 13–18 position.
Similar content being viewed by others
References
W. R. White and C. R. Noller, J. Am. Chem. Soc.,61, 983, 1939.
R. N. Jones, D. Todd, and C. R. Noller, J. Am. Chem. Soc.,61, 242, 1939.
J. E. Carson, D. B. Cosulich, and C. R. Noller, J. Am. Chem. Soc.,66, 1265, 1945.
P. Bilham and G. A. R. Kon, J. Chem. Soc., 1469, 1940.
F. A. Alves and C. R. Noller, J. Am. Chem. Soc.,74, 4043, 1952.
C. Djerassi, J. Osiecki, and W. Closson, J. Am. Chem. Soc.,81, 4587, 1959.
M. S. Newman, Steric Effects in Organic Chemistry [Russian translation], Moscow, 358, 1960.
E. L. Eliel, Stereochemistry of Carbon Compounds [Russian translation], Moscow, 201–203, 274–277, 1955.
C. Djerassi, G. H. Thomas, and H. Monsimer, J. Am. Chem. Soc.,77, 3579, 1955.
A. K. Barua and S. P. Raman, Tetrah.,7, 19, 1959.
A. K. Barua and S. P. Raman, Tetrah.,18, 155, 1962.
D. H. R. Barton, A. Hameed, and J. F. McGhie, J. Chem. Soc., 5179, 1962.
R. O. Dorchai, and J. B. Thompson, Tetrah. Let., 2223, 1965.
W. Jachimzyk and Z. Kasprzyk, Roczn. Chem.,36, 1615, 1962.
Author information
Authors and Affiliations
Additional information
Khimiya Prirodnykh Soedinenii, Vol. 3, No. 4, pp. 230–235, 1967
Rights and permissions
About this article
Cite this article
Belous, V.N. Decarboxylation products of echinocystic acid. Chem Nat Compd 3, 192–195 (1967). https://doi.org/10.1007/BF00564112
Issue Date:
DOI: https://doi.org/10.1007/BF00564112