Abstract
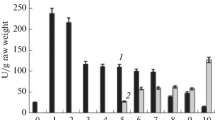
Malate dehydrogenase (MDH) band relative mobility (R m) and activity were examined in leaf extracts of Durrant's flax genotrophs, L and S, and flax genotypes, R and M. MDH activity in leaves from just below the inflorescence was higher in the two smaller, sparsely branched plant types, S and M, than in the larger, more branched plant types, L and R. The MDH electrophoretic banding pattern in flax leaf extracts consisted of three major anionic bands, MDH-1, MDH-2, and MDH-3. NoR m differences were detected between corresponding isozymes of genotypes R and M. For the genotrophs, however, all three bands of S migrated faster than the corresponding bands of L. Codominance was absent in F1 hybrids; SR m was dominant for MDH-2 and MDH-3 and LR m was dominant for MDH-1. The observations suggest that MDHR m in L and S may be controlled by a modifier locus (or loci). Previous studies indicate that a modifier locus may also control heritable genotrophic differences in peroxidase (PER) and acid phosphates (AP)R m. The three enzyme systems are compared.
Similar content being viewed by others
References
Bergmeyer, U. H., and Berndt, E. (1963). Malic dehydrogenase. In Bergmeyer, U. H. (eds.),Methods in Enzymatic Analysis Bergmeyer, Academic Press, New York, p. 757.
Durrant, A. (1971). Induction and growth of flax genotrophs.Heredity 27277.
Fieldes, M. A., and Tyson, H. (1984). Possible posttranslational modification, and its genetic control, in flax genotroph isozymes.Biochem. Genet. 2299.
Fieldes, M. A., Starobin, J., and Tyson, H. (1984). Visualization of both peroxidase and acid phosphatase isozymes from flax (Linum) on the same gels.Anal. Biochem. 137146.
Finnerty, V., and Johnson, G. (1979). Post-translational modification as a potential explanation of high levels of enzyme polymorphism: Xanthine dehydrogenase and alcohol oxidase inDrosophila melanogaster.Genetics 91695.
Gaudreault, P.-R., and Tyson, H. (1988). Elimination of differences in the mobility of flax isoperoxidases on PAGE by digestion with α-mannosidase.Plant Physiol. 86288.
Goodman, M. M., Stuber, C. W., Lee, C.-N., and Johnson, F. M. (1980). Genetic control of malate dehydrogenase isozymes in maize.Genetics 94153.
Grisebach, H. (1981). Lignins. In Conn, E. E. (ed.),The Biochemistry of Plants, Vol. 7, Secondary Plant Products Academic Press, New York, p. 457.
Harry, D. E. (1983). Identification of a locus modifying the electrophoretic mobility of malate dehydrogenase isozymes in incense-cedar(Calocedrus decurrens) and its implications for population studies.Biochem. Genet. 21417.
Hemmings, B. A., Zubenko, G. S., Haslik, A., and Jones, E. W. (1981). Mutant defective in processing of an enzyme located in the lysosome-like vacuole ofSaccharamyces cerevisiae.Proc. Natl. Acad. Sci. USA 78435.
Mader, M., and Füssl, R. (1982). Role of peroxidase in lignification of tobacco cells. II. Regulation by phenolic compounds.Plant Physiol. 701132.
Maehly, A. C., and Chance, B. (1954).Methods of Biochemical Analysis, Vol. 1 Interscience, New York, Part X, pp. 358–424.
McNaughton, S. J. (1974). Natural selection at the enzyme level.Am. Nat. 108616.
Pitel, J. A., and Cheliak, W. M. (1985). Methods to extract NAD+-malate dehydrogenase efficiently from white spruce needles.Physiol. Plant. 65129.
Shaw, G. R., and Prasad, R. (1970). Starch gel electrophoresis of enzymes: A compilation of recipes.Biochem. Genet. 4297.
Simon, J.-P. (1979a). Differences in thermal properties of NAD malate dehydrogenases in genotypes ofLathyrus japonicus Willd. (Leguminosae) from maritime and continental sites.Plant Cell Environ. 223.
Simon, J.-P. (1979b). Adaptation and acclimation of higher palnts at the enzyme level: Speed of acclimation for apparent energy of activation of NAD malate dehydrogenase inLathyrus japonicus Willd. (leguminosae).Plant Cell Environ. 235.
Tyson, H., Fieldes, M. A., Cheung, C., and Starobin, J. (1985). Isozyme relative mobility (R m) changes related to leaf position; Apparent smoothR m trends and some implications.Biochem. Genet. 23641.
Tyson, H., Fieldes, M. A., and Starobin, J. (1986). Genetic control of acid phosphataseR m and its relation to control of peroxidaseR m in flax(Linum) genotrophs.Biochem. Genet. 24369.
Welinder, K. G. (1976). Covalent structure of the glcoprotein horseradish peroxidase. (E.C. 1.11.1.7.).FEBS Lett. 7219.
Author information
Authors and Affiliations
Additional information
The financial assistance of the Natural Sciences and Engineering Research Council of Canada is acknowledged with thanks.
Rights and permissions
About this article
Cite this article
Fieldes, M.A. Relative mobility and activity of leaf malate dehydrogenase in flax(Linum usitatissimum) genotrophs and genotypes. Biochem Genet 26, 261–275 (1988). https://doi.org/10.1007/BF00561465
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00561465