Abstract
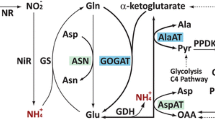
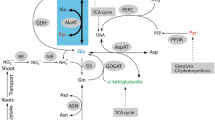
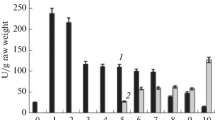
Ferguson plots demonstrated that corresponding malate dehydrogenase (MDH) isozymes of Durrant's L and S flax genotrophs differ in apparent molecular weight (MW) and also in net negative charge. The MW differences explain heritable differences in electrophoretic relative mobility (R m) between corresponding L and S isozymes. The MW for each MDH isozyme was higher for L than for S and resulted in a slowerR m for L. The net negative charge for each isozyme was higher for L than for S. MDH isozymes also differ in MW within L and S. MW was lower for isozymes in leaves from the bottom of the stem than in leaves from the top of the stem, particularly in L. Integration of information on the MDH isozyme system in the flax genotrophs and information on the peroxidase system suggests the possibility that common modifier loci may controlR m in both enzymes.
Similar content being viewed by others
References
Cassman, M., and King, R. (1972). Molecular weight studies on the supernatant malate dehydrogenase isolated from beef heart: Evidence for a protyolytic contaminant in the purified preparation.Biochim. Biophys. Acta 257143.
Durrant, A. (1971). Induction and growth of flax genotrophs.Heredity 27277.
Ferguson, K. A. (1964). Starch-gel electrophoresis—application to the classification of pituitary proteins and polypeptides.Metabolism 13985.
Fieldes, M. A. (1988). Relative mobility and activity of leaf malate dehydrogenase in flax (Linum usitatissimum) genotrophs and genotypes.Biochem. Genet. 26261.
Fieldes, M. A., and Gray, T. J. (1988). Rm differences in leaf malate dehydrogenases of flax (Linum usitatissimum) genotrophs: Apparent developmental effects.J. Exp. Bot. 39.
Fieldes, M. A., and Tyson, H. (1984). Possible posttranslational modification, and its genetic control, in flax genotroph isozymes.Biochem. Genet. 2299.
Fieldes, M. A., Deal, C. L., and Tyson, H. (1977). Preliminary characterization of peroxidase isozymes isolated from two flax genotrophs.Can. J. Bot. 551465.
Fieldes, M. A., Starobin, J., and Tyson, H. (1984). Visualization of both peroxidate and acid phosphatase isozymes from flax (Linum) on the same gels.Anal. Biochem. 137146.
Finnerty, V., and Johnson, G. (1979). Post-translational modification as a potential explanation of high levels of enzyme polymorphism: Xanthine dehydrogenase and alcohol oxidase inDrosophila melanogaster.Genetics 91695.
Gonenne, A., and Lebowitz, J. (1975). Estimation of molecular weights of small proteins on polyacrlamide gel electrophoresis.Anal. Biochem. 64414.
Goodman, M. M., Stuber, C. W., Lee, C. -N., and Johnson, F. M. (1980). Genetic control of malate dehydrogenase isozymes in maize.Genetics 94153.
Harry, D. E. (1983). Identification of a locus modifying the electrophoretic mobility of malate dehydrogenase isozymes in incense-cedar (Calocedrus decurrens) and its implications for population studies.Biochem. Genet. 21417.
Hemmings, B. A., Zubenko, G. S., Haslik, A., and Jones, E. W. (1981). Mutant defective in processing of an enzyme located in the lysosome-like vacuole ofSaccharamyces cerevisiae.Proc. Natl. Acad. Sci. USA 78435.
Newton, K. J., and Schwartz, D. (1980). Genetic basis of the major malate dehydrogenase isozymes in maize.Genetics 95425.
Rodbard, D., and Chrambach, A. (1974). Quantitative polyacrylamide gel electrophoresis: Mathematical and statistical analysis of data. In Allen, R. C., and Maurer, H. R. (eds.),Electrophoresis and Isoelectric Focusing in Polyacrylamide Gel de Gruyter, Berlin, pp. 28–65.
Shaw, G. R., and Prasad, R. (1970). Starch gel electrophoresis of enzymes: A compilation of recipes.Biochem. Genet. 4297.
Ting, I. P. (1968). Malic dehydrogenase in corn root tips.Arch. Biochem. Biophys. 1261.
Ting, I. P., Fuhr, I., Curry, R., and Zschoche, W. C. (1975). Malate dehydrogenase isozymes in plants: Preparation, properties, and biological significance. In Market, C. L. (ed.),Isozymes, Vol. II. Physiological Function Academic Press, New York, pp. 369–384.
Tyson, H., and Fieldes, M. A. (1982). Molecular weight and net charge of peroxidase isozymes in F1 hybrids between L and S flax genotrophs.Biochem. Genet. 20919.
Tyson, H., Fieldes, M. A., Cheung, C., and Starobin, J. (1985). Isozyme relative mobility (R m) changes related to leaf position; Apparent smoothR m trends and some implications.Biochem. Genet. 23641.
Tyson, H., Fieldes, M. A., and Starobin, J. (1986). Genetic control of acid phosphatase Rm and its relation to control of peroxidaseR m in flax (Linum) genotrophs.Biochem. Genet. 24369.
van den Berg, B. M., and Wijsman, H. J. W. (1981). Genetics of the peroxidase isoenzymes in petunia. 1. Organ specificity ang general genetic aspects of the peroxidase isoenzymes.Theor. Appl. Genet. 6071.
Author information
Authors and Affiliations
Additional information
The financial assistance of the Natural Sciences and Engineering Research Council of Canada is acknowledged with thanks.
Rights and permissions
About this article
Cite this article
Fieldes, M.A., Dixon, B. Malate dehydrogenase isozymes in flax genotroph leaves: Differences in apparent molecular weight and charge between and within L and S. Biochem Genet 26, 249–260 (1988). https://doi.org/10.1007/BF00561464
Received:
Revised:
Issue Date:
DOI: https://doi.org/10.1007/BF00561464