Abstract
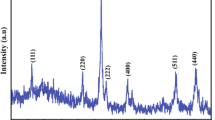
Ferric chloride solution was neutralized using sodium hydroxide as the precipitant to the extent that the final pH of the suspension was ∼7. After thorough washing the precipitated gel was dried in air at 60° C for 24 h to yield brown ferric oxide hydrate xerogel. The xerogel was examined at room temperature using infrared (IR) spectroscopy, powder X-ray diffraction (XRD), electron optical measurements, magnetometry and Mössbauer resonance spectroscopy. It has been concluded that the structural, morphological and magnetic characteristics of the xerogel are identifiable with those of the protoferrihydrite phase.
Similar content being viewed by others
References
H. B. Weiser and W. O. Milligan, in “Advances in Colloid Science”, Vol. I, edited by E. O. Kramer, E. F. Bartet and S. S. Kistler (Interscience, New York, 1942) p. 227.
Idem, J. Phys. Chem. 44 (1940) 1081.
R. C. Mackenzie and G. Berggren, in “Differentiatial Thermal Analysis”, Vol. I, edited by R. C. Mackenzie (Academic Press, London, 1970) p. 277.
R. C. Mackenzie and R. C. Meldau, Mineral. Mag. 33 (1959) 153.
M. Kobayashi and M. Uda, J. Non Crystalline Solids 29 (1978) 419.
M. Magini, J. Inorg. Nucl. Chem. 39 (1977) 409.
A. A. Van Der Giessen, Philips Res. Rep. Supl. 12 (1968.
Idem, J. Inorg. Nucl. Chem. 28 (1966) 2155.
S. Okamoto, H. Sekizawa and S. I. Okamoto, in Proceedings 7th International Conference on the Reactivity of Solids, edited by J. S. Anderson, M. W. Roberts and F. S. Stone (Chapman and Hall, London, 1972) p. 341.
K. M. Towe and W. F. Bradley, J. Coll. Interface Sci. 24 (1967) 384.
F. V. Chukhrov, B. B. Zvygin, A. I. Gorshkov, L. P. Ermilova and E. S. Rudnitskaya, Izvestia. AN SSSR Ser. geol. 1 (1971) 3.
F. V. Chukhrov, B. B. Zvyagin, L. P. Ermilova and A. I. Gorshkov, in Proceedings International Clay Conference, Madrid, edited by J. M. Serratsova (Sociedad Espanola de Arcillas and Association Internationale pour L'Etude des Argilles, 1972) p. 397.
M. H. Hey and P. G. Embrey, Mineral. Mag. 39 (1974) 903.
M. Fleischer, C. Y. Chao and A. Kato, Amer. Mineral. 60 (1975) 485.
T. A. Jackson and W. D. Keller, Amer. J. Sci. 269 (1970) 446.
U. Schwertmann and W. R. Fischer, Geoderma 10 (1973) 237.
U. Schwertmann, D. G. Schulze and E. Murad, Soil Sci. Soc. Am. J. 46 (1982).
E. Murad and U. Schwertmann, Amer. Mineral. 65 (1980) 1044.
A. C. Vajpei, PhD thesis, University of Roorkee, Roorkee, India (1978).
I. P. Saraswat, A. C. Vajpei, V. K. Garg, V. K. Sharma and Nam Prakash, J. Coll. Interface Sci. 73 (1980) 373.
I. P. Saraswat, A. C. Vajpei, V. K. Garg, Indian J. Chem. 15A (1977) 493.
Y. I. Ryskin, in “The IR spectra of Minerals”, edited by V. C. Farmer (Mineralogical Society, London, 1974) p. 137.
R. A. Nyquist and R. O. Kagel, “Infrared Spectra of Inorganic Compounds” (Academic Press, New York, 1971).
C. N. R. Rao, ‘Chemical Applications of Infrared Spectroscopy” (Academic Press, New York, 1963).
F. F. Bentley, L. D. Smithson and A. L. Rozeck, “Infrared Spectra and Characteristic Frequencies 700–300 cm−1” (Interscience, New York, 1968).
K. Kauffman and F. Hazel, J. Inorg. Nucl. Chem. 37 (1975) 1139.
J. D. Russel, Clay Minerals 14 (1979) 109.
G. Matzen and P. Poix, J. Mater. Sci. 17 (1982) 701.
C. W. Childs and J. H. Johnston, Aust. J. Soil. Res. 18 (1980) 245.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Vajpei, A.C., Uma, Saraswat, I.P. et al. Characterization of brown ferric oxide hydrate xerogel. J Mater Sci 19, 3341–3346 (1984). https://doi.org/10.1007/BF00549824
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF00549824